- +86-0533-2185556
- WhatsApp: +86 15965530500
- admin@hangyubiotech.com
Your Location:Home >Products >Pharmaceutical intermediates >4651-91-6
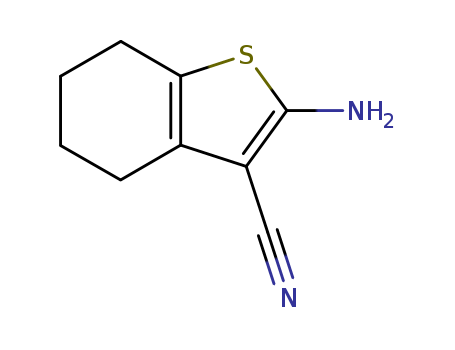

pd_meltingpoint:151-153 °C
Purity:99%
InChI:InChI=1/C9H10N2S/c10-5-7-6-3-1-2-4-8(6)12-9(7)11/h1-4,11H2
Dual targeting of epidermal growth facto...
A string of novel heterocyclic mono azo ...
The reaction of 2-amino-3-cyano-tetrahyd...
Two series of thieno[2,3-d][1,2,3]triazi...
The synthesis of a variety of novel 5-su...
Liver cancer is one of the most importan...
In this present study, a series of novel...
The discovery and development of isoform...
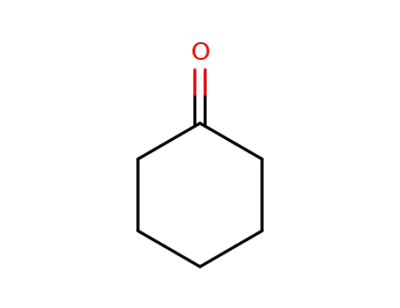
cyclohexanone

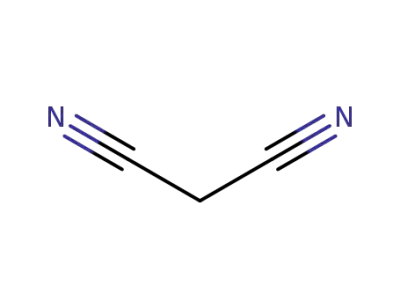
malononitrile

![3-cyano-2-amino-4,5,6,7-tetrahydrobenzo[b]thiophene](/upload/2026/5/62e4581a-1507-430d-b8a9-9f7e7607337c.png)
3-cyano-2-amino-4,5,6,7-tetrahydrobenzo[b]thiophene
| Conditions | Yield |
|---|---|
|
With
octasulfur; bovine serum albumin;
In
N,N-dimethyl-formamide;
at 50 ℃;
for 4h;
Reagent/catalyst;
Solvent;
Time;
Temperature;
Concentration;
Catalytic behavior;
|
99% |
|
With
sulfur; L-proline;
In
N,N-dimethyl-formamide;
at 60 ℃;
for 10h;
|
97% |
|
With
morpholine; sulfur;
In
ethanol;
at 30 ℃;
for 8h;
|
97% |
|
With
sulfur; diethylamine;
In
water;
for 0.0833333h;
Sonication;
|
96% |
|
With
sulfur;
In
neat (no solvent);
at 100 ℃;
for 4h;
|
96% |
|
With
sulfur;
at 100 ℃;
for 0.0833333h;
Reagent/catalyst;
Catalytic behavior;
Green chemistry;
|
95% |
|
With
potassium carbonate; sulfur;
In
ethanol;
at 85 ℃;
for 0.25h;
under 10343.2 Torr;
Microwave irradiation;
|
95% |
|
With
sulfur; diethylamine;
In
ethanol;
at 70 ℃;
for 17h;
|
94% |
|
With
sulfur; potassium carbonate;
In
ethanol;
for 3h;
Reflux;
|
94% |
|
With
piperidine; sulfur;
In
methanol;
at 30 ℃;
for 1h;
|
94.9% |
|
With
sulfur;
potassium fluoride on basic alumina;
In
ethanol;
at 100 ℃;
for 0.1h;
under 12928.7 Torr;
microwave irradiation;
|
92% |
|
With
sulfur;
In
ethanol;
at 50 ℃;
for 1h;
|
92% |
|
With
morpholine; sulfur;
In
ethanol;
Reflux;
|
92% |
|
With
sulfur; calcined Mg-Al hydrotalcite;
In
ethanol;
at 60 ℃;
for 8h;
|
91% |
|
With
sulfur;
In
ethanol;
at 78 ℃;
for 1.08333h;
|
90% |
|
With
sulfur;
at 100 ℃;
for 2h;
Reagent/catalyst;
Green chemistry;
|
90% |
|
With
caesium carbonate; sulfur;
In
ethanol;
for 2.33333h;
Reflux;
|
89% |
|
With
sulfur;
In
ethanol;
for 4h;
Reflux;
|
89% |
|
With
sulfur; triethylamine;
In
water;
at 20 ℃;
for 6h;
|
89% |
|
With
morpholine; sulfur;
In
ethanol;
at 50 - 75 ℃;
for 10h;
|
89% |
|
With
sulfur; diethylamine;
In
neat (no solvent);
for 0.333333h;
Reagent/catalyst;
Time;
Milling;
|
88% |
|
With
sulfur;
at 60 ℃;
for 2h;
Ionic liquid;
Green chemistry;
|
88% |
|
With
sulfur; choline chloride; urea; sodium hydroxide;
In
water;
at 60 ℃;
Green chemistry;
|
88% |
|
With
sulfur;
In
neat (no solvent);
at 60 ℃;
for 2h;
Reagent/catalyst;
Time;
Catalytic behavior;
|
88% |
|
With
sulfur; diethylamine;
In
ethanol;
at 40 - 50 ℃;
for 2h;
|
84% |
|
cyclohexanone; malononitrile;
In
water;
for 0.333333h;
Sonication;
Green chemistry;
With
Na2S6;
In
water;
for 0.5h;
Reagent/catalyst;
Solvent;
Sonication;
Green chemistry;
|
84% |
|
With
zinc(II) oxide; sulfur;
In
neat (no solvent);
at 100 ℃;
for 6h;
Concentration;
Time;
|
81% |
|
With
zinc ferrite; sulfur;
In
neat (no solvent);
at 100 ℃;
for 4h;
Catalytic behavior;
Green chemistry;
|
80% |
|
With
morpholine; sulfur;
In
ethanol;
at 45 - 60 ℃;
for 18.25h;
|
80% |
|
With
sodium aluminate; sulfur;
In
ethanol;
at 60 ℃;
for 7h;
|
80% |
|
cyclohexanone; malononitrile;
With
diethylamine;
In
ethanol;
for 0.166667h;
Reflux;
With
sulfur;
In
ethanol;
for 3h;
Reflux;
|
75% |
|
cyclohexanone; malononitrile;
With
sulfur;
In
ethanol;
at 20 ℃;
for 0.166667h;
With
morpholine;
at 60 ℃;
for 12h;
|
62.8% |
|
With
morpholine; sulfur;
In
ethanol;
for 2h;
Reflux;
|
57% |
|
cyclohexanone; malononitrile;
With
sulfur;
In
ethanol;
for 0.0833333h;
With
morpholine;
In
ethanol;
at 20 - 60 ℃;
for 5.5h;
|
56% |
|
With
morpholine; cyanoacetic acid tert-butyl ester; sulfur;
In
ethanol;
at 40 ℃;
for 20h;
|
51% |
|
With
sulfur; triethylamine;
In
ethanol;
for 16h;
Reflux;
|
44% |
|
With
sulfur; triethylamine;
In
ethanol;
for 16h;
Reflux;
|
44% |
|
With
N-ethylmorpholine;; sulfur;
In
ethanol;
at 20 ℃;
for 3h;
|
33% |
|
With
sulfur; diethylamine;
In
ethanol;
|
|
|
With
sulfur; diethylamine;
In
ethanol;
at 20 ℃;
|
|
|
With
morpholine; sulfur;
In
ethanol;
at 0 - 20 ℃;
|
|
|
With
morpholine; sulfur;
In
ethanol;
for 2h;
Heating;
|
|
|
With
sulfur; triethylamine;
In
ethanol;
for 6h;
Heating / reflux;
|
|
|
With
sulfur; diethylamine;
In
ethanol;
|
|
|
With
morpholine; sulfur;
In
ethanol;
Reflux;
|
|
|
With
sulfur;
|
|
|
With
sulfur; diethylamine;
In
ethanol;
at 20 - 70 ℃;
|
|
|
With
sulfur; triethylamine;
|
|
|
With
morpholine; sulfur;
In
ethanol;
Reflux;
|
|
|
With
morpholine; sulfur;
|
|
|
With
sulfur; diethylamine;
|
|
|
With
morpholine; sulfur;
at 20 ℃;
|
|
|
With
morpholine; sulfur;
In
ethanol;
at 20 ℃;
|
|
|
With
morpholine; sulfur;
In
ethanol;
at 60 ℃;
for 12h;
|
|
|
With
morpholine; sulfur;
In
ethanol;
at 20 ℃;
|
|
|
With
morpholine; sulfur;
In
ethanol;
at 5 - 10 ℃;
for 3h;
|
|
|
With
sulfur; sodium hydroxide;
In
tert-butyl alcohol;
at 20 ℃;
for 0.5h;
Reagent/catalyst;
Solvent;
Inert atmosphere;
|
94 %Chromat. |
|
With
ethanol; sulfur; triethylamine;
for 0.5h;
Reflux;
|
|
|
With
sulfur; triethylamine;
In
ethanol;
Reflux;
|
|
|
With
morpholine; sulfur;
In
ethanol;
at 5 - 10 ℃;
for 3h;
|
|
|
With
hydrogen sulfide; diethylamine;
|
|
|
With
sulfur; triethylamine;
In
ethanol;
|
|
|
With
morpholine; sulfur;
In
butan-1-ol;
at 150 ℃;
for 0.166667h;
Cooling with ice;
Microwave irradiation;
|
|
|
With
morpholine; sulfur;
In
butan-1-ol;
at 150 ℃;
for 0.166667h;
Microwave irradiation;
|
|
|
With
morpholine; sulfur;
In
ethanol;
|
|
|
With
morpholine; sulfur;
In
ethanol;
at 60 ℃;
for 24h;
|
|
|
With
sulfur; diethylamine;
In
methanol;
|
|
|
With
morpholine; sulfur;
In
ethanol;
|
|
|
With
piperidine; sulfur;
In
ethanol;
at 50 ℃;
for 2h;
|
|
|
With
sulfur;
In
ethanol;
for 4h;
Reflux;
|
|
|
With
sulfur;
In
ethanol;
|
|
|
With
morpholine; sulfur;
In
ethanol;
at 5 - 20 ℃;
for 1h;
|
|
|
With
sulfur;
In
ethanol;
at 60 ℃;
for 1.5h;
|
|
|
With
sulfur;
In
ethanol;
at 60 ℃;
|
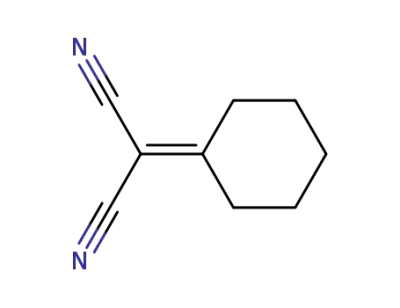
2-(cyclohexylidene)malononitrile

![3-cyano-2-amino-4,5,6,7-tetrahydrobenzo[b]thiophene](/upload/2026/5/62e4581a-1507-430d-b8a9-9f7e7607337c.png)
3-cyano-2-amino-4,5,6,7-tetrahydrobenzo[b]thiophene
| Conditions | Yield |
|---|---|
|
With
sulfur; tetraethylammonium hexafluorophosphate; acetonitrile;
at 20 ℃;
Electrolysis;
Inert atmosphere;
|
99% |
|
With
sulfur; L-proline;
In
dimethyl sulfoxide;
at 75 ℃;
|
74% |
|
With
sulfur; ethylenediamine diacetic acid;
|
|
|
With
morpholine; sulfur;
In
N,N-dimethyl-formamide;
at 20 - 120 ℃;
for 0.166667h;
|
|
|
With
1,4-diaza-bicyclo[2.2.2]octane; sulfur;
In
water;
at 20 ℃;
for 7h;
|

cyclohexanone

malononitrile

2-(cyclohexylidene)malononitrile
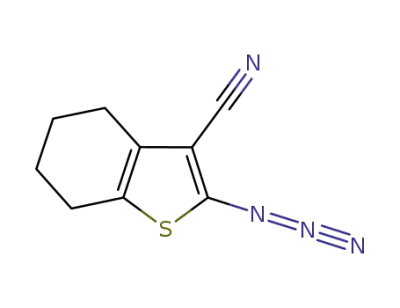
2-azido-4,5,6,7-tetrahydrobenzo[b]thiophene-3-carbonitrile
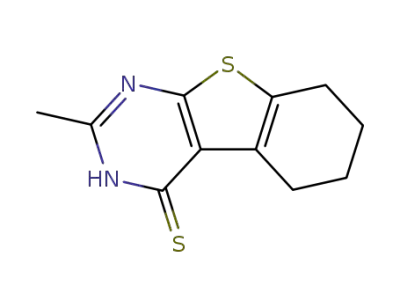
2-methyl-5,6,7,8-tetrahydro-3H-benzo[4,5]thieno[2,3-d]pyrimidine-4-thione
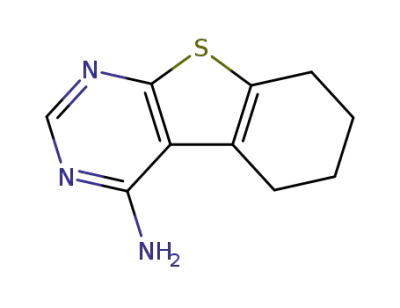
5,6,7,8-tetrahydrobenzo[b]thiopheno[2,3-d]pyrimidin-4-ylamine
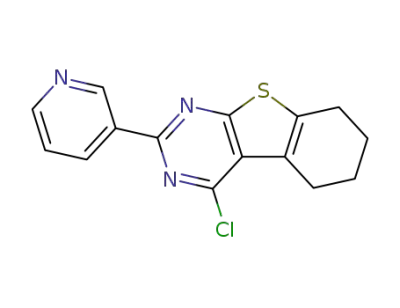
4-chloro-2-(pyridin-3-yl)-5,6,7,8-tetrahydro-[1]-benzothieno[2,3-d]pyrimidine
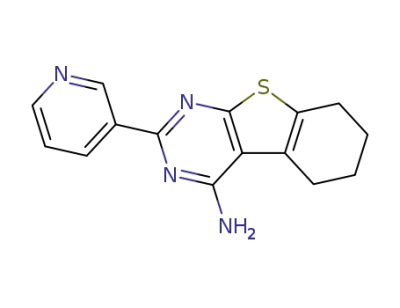
2-(pyridin-3-yl)-5,6,7,8-tetrahydrobenzo[4,5]thieno[2,3-d]pyrimidin-4-amine
CAS:115473-15-9
CAS:1173-88-2
CAS:71320-77-9
CAS:516-54-1