- +86-0533-2185556
- WhatsApp: +86 15965530500
- admin@hangyubiotech.com
Your Location:Home >Products >Nucleosides >58699-61-9
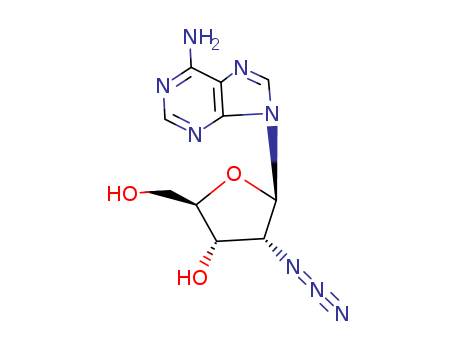

Purity:99%
|
General Description |
2'-Azido-d-adenosine is a chemical compound consisting of an adenosine molecule with an azido group attached at the 2' position. It is a modified form of the natural nucleoside adenosine, and it is commonly used in chemical biology and biochemistry research. The azido group makes the compound reactive and is often used in studies of nucleic acid structure and function, as well as in labeling and tagging experiments to track and study the behavior of adenosine and related molecules. 2'-Azido-d-adenosine has also been investigated for its potential therapeutic applications, particularly in the development of antiviral drugs and cancer treatments. Due to its unique properties, this compound has shown promise in both research and potential medical uses. |
InChI:InChI=1/C10H13N8O3/c11-8-6-9(14-2-13-8)18(3-15-6)10-5(16-17-12)7(20)4(1-19)21-10/h2-5,7,10,12,19-20H,1H2,(H2,11,13,14)/q+1
Non-hydrolyzable substrate analogs for t...
Olygoadenylic acids containing 2'-5' pho...
-
-
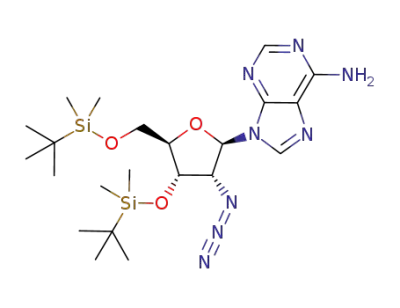
9-((2R,3R,4S,5R)-3-azido-4-((tert-butyldimethylsilyl)oxy)-5-(((tert-butyldimethylsilyl)oxy)methyl)tetrahydrofuran-2-yl)-9H-purin-6-amine

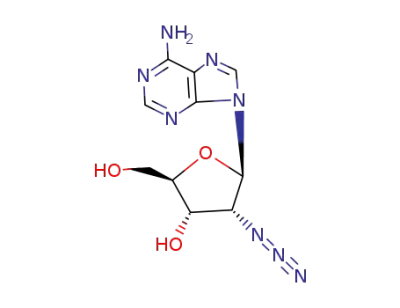
2'-Azido-2'-deoxyadenosine
| Conditions | Yield |
|---|---|
|
With
ammonium fluoride;
In
methanol;
at 60 ℃;
Inert atmosphere;
|
91% |
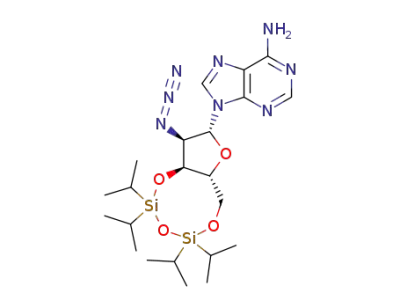
2'?azido?2'?deoxy?3',5'?O?(1,1,3,3?tetraisopropyldisiloxane?1,3?diyl)adenosine


2'-Azido-2'-deoxyadenosine
| Conditions | Yield |
|---|---|
|
With
tetrabutyl ammonium fluoride;
In
tetrahydrofuran;
for 0.166667h;
Ambient temperature;
|
84% |
|
With
tetrabutyl ammonium fluoride;
Yield given;
|

2'?azido?2'?deoxy?3',5'?O?(1,1,3,3?tetraisopropyldisiloxane?1,3?diyl)adenosine
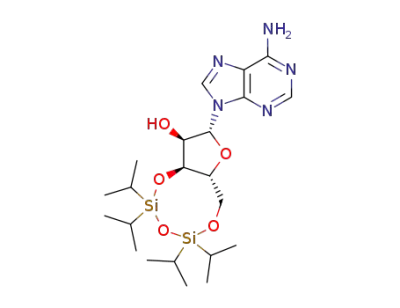
3',5'-O-(1,1,3,3-tetra-isopropyldisiloxane-1,3-diyl)adenosine
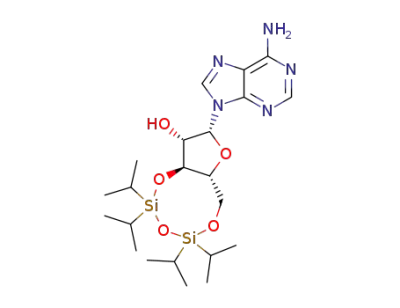
9-[3',5'-O-(1,1,3,3-tetraisopropyldisiloxane-1,3-diyl)-1-β-D-arabinofuranosyl]adenine
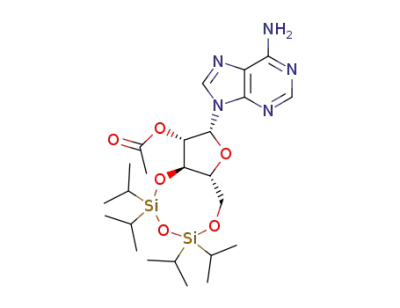
Acetic acid (2R,3S,3aR,9aR)-2-(6-amino-purin-9-yl)-5,5,7,7-tetraisopropyl-tetrahydro-1,4,6,8-tetraoxa-5,7-disila-cyclopentacycloocten-3-yl ester
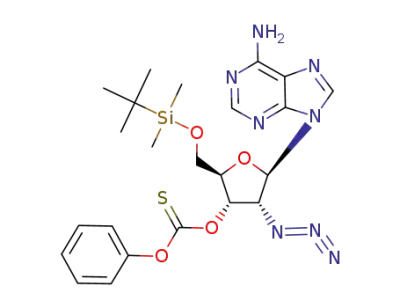
Thiocarbonic acid O-[(2R,3S,4R,5R)-5-(6-amino-purin-9-yl)-4-azido-2-(tert-butyl-dimethyl-silanyloxymethyl)-tetrahydro-furan-3-yl] ester O-phenyl ester
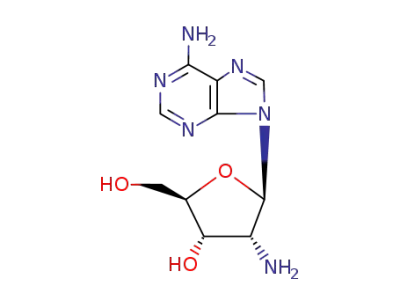
2'-amino-2'-deoxyadenosine
CAS:112163-33-4
CAS:112-84-5
CAS:18835-32-0
CAS:4382-63-2