- +86-0533-2185556
- WhatsApp: +86 15965530500
- admin@hangyubiotech.com
Your Location:Home >Products >API >43120-28-1

pd_meltingpoint:162-163℃
Purity:99%
InChI:InChI=1/C9H8N2O2/c1-13-9(12)8-6-4-2-3-5-7(6)10-11-8/h2-5H,1H3,(H,10,11)
The indazole scaffold represents a promi...
The invention relates to preparation met...
A novel universalN-transfer reagent for ...
Disclosed herein are compounds of formul...
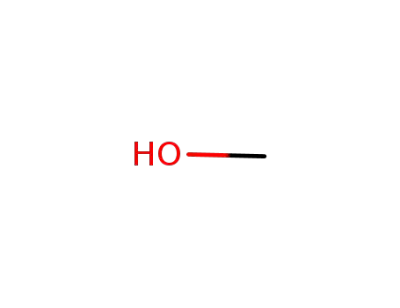
methanol

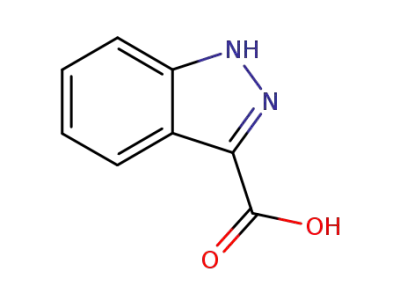
1H-Indazole-3-carboxylic acid

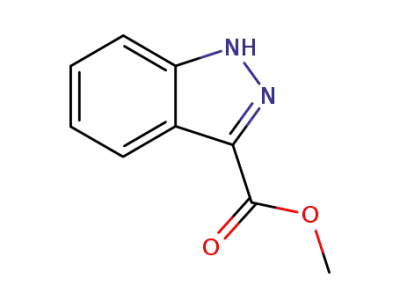
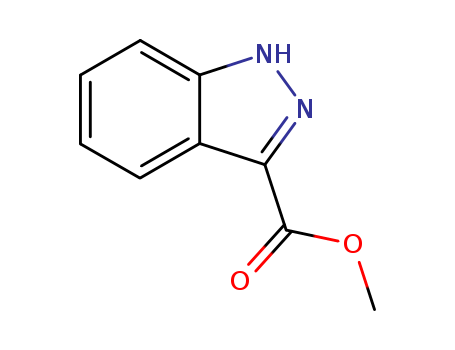
methyl 1H-indazole-3-carboxylate
| Conditions | Yield |
|---|---|
|
With
sulfuric acid;
for 2h;
Heating;
|
98% |
|
With
sulfuric acid;
for 2h;
Reflux;
|
95% |
|
With
thionyl chloride;
for 3h;
Reflux;
|
95% |
|
With
thionyl chloride;
at 0 ℃;
for 1.5h;
Reflux;
|
94% |
|
With
thionyl chloride;
at 0 - 20 ℃;
|
93% |
|
With
thionyl chloride;
at 20 ℃;
Cooling with ice;
|
93% |
|
With
thionyl chloride;
at 0 ℃;
for 5h;
Reflux;
|
92% |
|
With
sulfuric acid;
for 16h;
Reflux;
|
92% |
|
methanol; 1H-Indazole-3-carboxylic acid;
With
sulfuric acid;
at 80 ℃;
for 16h;
With
sodium hydrogencarbonate;
In
water; ethyl acetate;
|
90.1% |
|
With
sulfuric acid;
for 24h;
Reflux;
|
88% |
|
methanol;
With
acetyl chloride;
for 0.166667h;
Cooling with ice;
1H-Indazole-3-carboxylic acid;
at 20 ℃;
|
85% |
|
With
sulfuric acid;
at 80 ℃;
|
82% |
|
methanol; 1H-Indazole-3-carboxylic acid;
With
sulfuric acid;
for 4h;
Reflux;
With
sodium hydrogencarbonate;
In
water; ethyl acetate;
|
78% |
|
With
sulfuric acid;
for 10h;
Heating / reflux;
|
48% |
|
With
sulfuric acid;
|
|
|
With
hydrogenchloride;
Heating;
|
|
|
sulfuric acid;
Heating / reflux;
|
|
|
With
sulfuric acid;
at 80 ℃;
for 24h;
|
|
|
With
sulfuric acid;
|
|
|
With
thionyl chloride;
at 20 ℃;
for 24h;
|
|
|
With
sulfuric acid;
for 20h;
Heating / reflux;
|
|
|
With
sulfuric acid;
In
water;
at 80 ℃;
for 24h;
|
|
|
With
hydrogenchloride;
In
1,4-dioxane;
at 20 ℃;
for 24h;
|
|
|
With
sulfuric acid;
Reflux;
|
|
|
With
sulfuric acid;
for 5h;
Reflux;
|
|
|
With
sulfuric acid;
at 60 ℃;
|
22.76 g |
|
With
sulfuric acid;
for 4h;
Reflux;
|
|
|
With
thionyl chloride;
at 0 - 80 ℃;
for 2h;
|
|
|
methanol;
With
acetyl chloride;
for 0.5h;
Cooling with ice;
1H-Indazole-3-carboxylic acid;
for 4h;
Reflux;
|
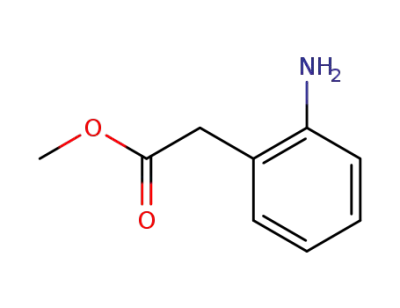
2-aminophenylacetic acid methyl ester


methyl 1H-indazole-3-carboxylate
| Conditions | Yield |
|---|---|
|
With
tert.-butylnitrite; acetic acid;
In
acetonitrile;
at 20 - 25 ℃;
for 0.5h;
Flow reactor;
|
92% |
|
Multi-step reaction with 2 steps
1: sodium nitrite / water; tetrahydrofuran / 0.25 h / -5 °C
2: potassium carbonate / water; tetrahydrofuran / 0.25 h / 0 - 20 °C
With
potassium carbonate; sodium nitrite;
In
tetrahydrofuran; water;
|

methanol

1H-Indazole-3-carboxylic acid
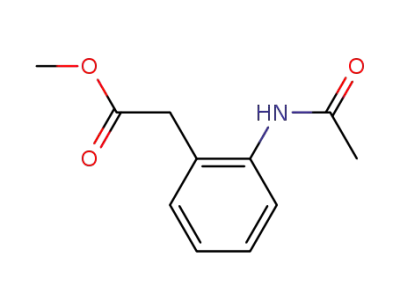
methyl 3-(2-acetamidophenyl)propanoate
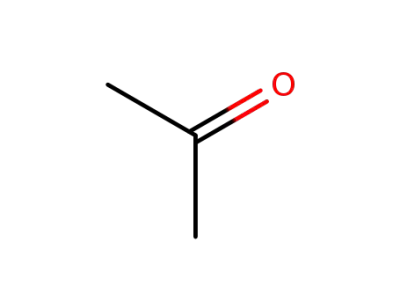
acetone
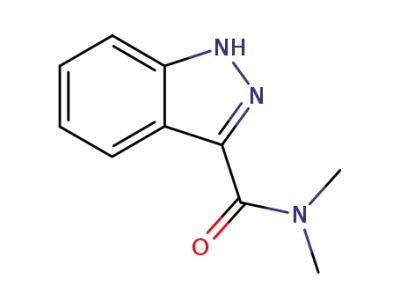
1(2)H-indazole-3-carboxylic acid dimethylamide
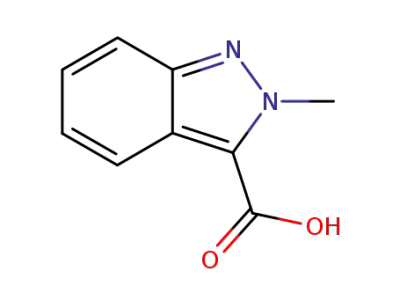
2-Methylindazole-3-carboxylic acid
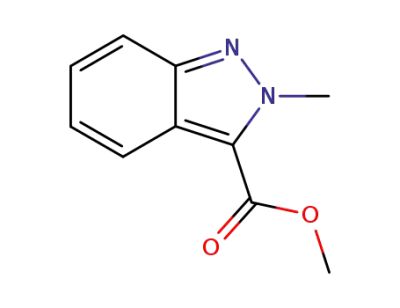
3-methoxycarbonyl-2-methylisoindazole
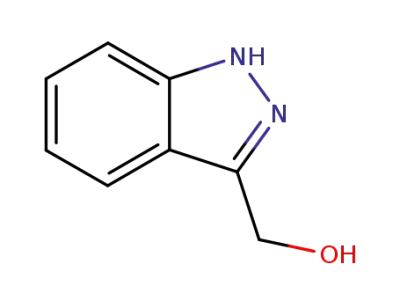
3-(hydroxymethyl)-1H-indazole
CAS:112163-33-4
CAS:112-84-5
CAS:9000-90-2
CAS:14172-91-9