- +86-0533-2185556
- WhatsApp: +86 15965530500
- admin@hangyubiotech.com
Your Location:Home >Products >API >14172-91-9
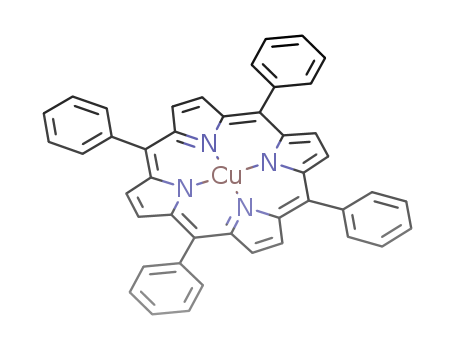

Purity:99%
|
General Description |
Copper TPP, also known as copper triphenylphosphine, is a coordination compound that contains a copper atom bonded to three triphenylphosphine ligands. It is used as a catalyst in organic synthesis reactions, particularly in the formation of carbon-carbon and carbon-heteroatom bonds. Copper TPP is known for its ability to promote C-C bond forming reactions, such as the coupling of aryl halides with terminal alkynes, and is widely utilized in the pharmaceutical and agrochemical industries for the synthesis of complex organic molecules. Additionally, copper TPP has been studied for its potential application in the development of new materials and nanotechnologies due to its unique electronic and optical properties. |
InChI:InChI=1/C44H30N4.Cu/c1-5-13-29(14-6-1)38-27-37-26-35-22-21-33(45-35)25-34-23-24-36(46-34)28-39-40(30-15-7-2-8-16-30)41(31-17-9-3-10-18-31)44(48-39)42(43(38)47-37)32-19-11-4-12-20-32;/h1-28,45,48H;/q;+2/b33-25-,34-25-,35-26-,36-28-,37-26-,39-28-,43-42-,44-42-;
-
A series of β-alkylaminoporphyrins conju...
Copper containing macromolecules, viz., ...
Chloro-form was found to occupy the latt...
Coordination of three azaporphines, name...
Acid–base and coordination properties of...
A non-covalent functionalization based o...
The oxidation of benzyl alcohols has bee...
In this work, two new single crystals of...
Porphyrins are known as unique ligands f...
Complexation between 5,10,15,20-tetraphe...
In the NMR and ESR time-scale of this ex...
Mixing Cu(II)Cl2 with free base tetraphe...
The metal-exchange reaction of cadmium 5...
The rate of incorporation of Cu(II) into...
The formation of the sitting-atop (SAT) ...
The reactions of tetraphenylporphine (H2...
Herein the synthesis and properties of t...
Metalloporphyrins are involved in many a...
The push-pull electronic and steric effe...
Electrochemical N2reduction to NH3is an ...
A novel and efficient protocol for oxida...
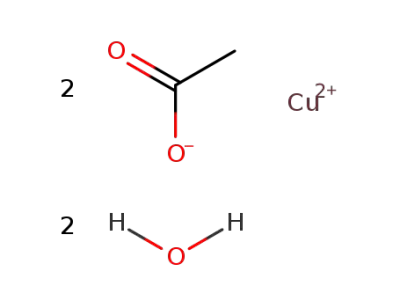
copper(II) acetate dihydrate

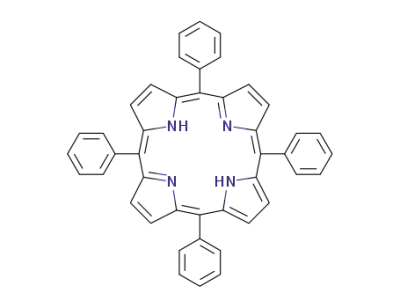
5,15,10,20-tetraphenylporphyrin

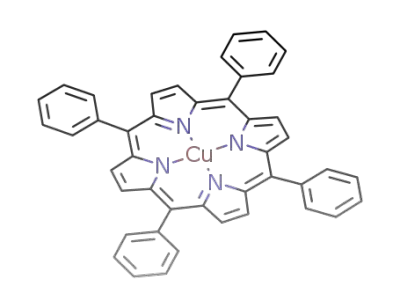
(tetraphenylporphyrin)copper(II)
| Conditions | Yield |
|---|---|
|
In
methanol; chloroform;
at 20 ℃;
|
99% |
|
With
silica gel;
for 0.25h;
Milling;
|
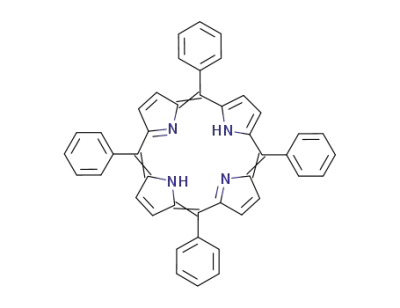
5,10,15,20-tetraphenyl-21H,23H-porphine

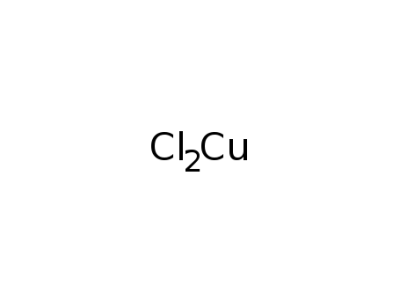
copper dichloride


(tetraphenylporphyrin)copper(II)
| Conditions | Yield |
|---|---|
|
In
solid;
High Pressure; 1000-6000 MPa at room temp.; extrd. (chloroform), chromy.(alumina); UV;
|
64% |
|
In
dimethyl sulfoxide;
byproducts: HCl; kinetic of react. of CuCl2 with H2TPP in DMSO studied at temp. 343-363 K; not isolated detn. by spectrophotometric method;
Kinetics;
|
|
|
In
ethanol; acetic acid;
byproducts: HCl; 290-308 K, various compn. of the binary solvents, hermetically sealed quartz cuvette; not isolated, detected by UV;
Kinetics;
|
|
|
With
LiCl;
In
ethanol; acetic acid;
byproducts: HCl; 298 K, 1:1 mixture of HAc and EtOH, hermetically sealed quartz cuvette; not isolated, detected by UV;
Kinetics;
|
|
|
In
pentan-1-ol; acetic acid;
byproducts: HCl; 288 - 308 K, various compn. of the binary solvents, hermetically sealed quartz cuvette; not isolated, detected by UV;
Kinetics;
|
|
|
In
acetic acid;
byproducts: HCl; 289 - 308 K, hermetically sealed quartz cuvette; not isolated, detected by UV;
Kinetics;
|
|
|
With
sodium carbonate;
In
water;
at 350 ℃;
for 0.0666667h;
Temperature;
Time;
Inert atmosphere;
Sealed tube;
|

5,10,15,20-tetraphenyl-21H,23H-porphine
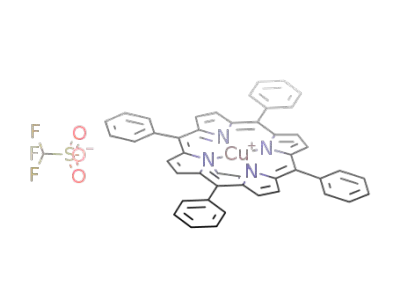
(N-ethyl-5,10,15,20-tetraphenylporphinato)copper(II) trifluoromethanesulfonate

dibutylamine

5,15,10,20-tetraphenylporphyrin
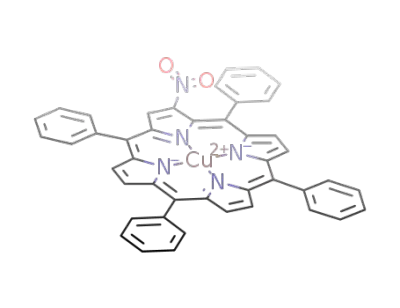
(2-nitro-5,10,15,20-tetraphenylporphyrinato)copper(II)
copper(II) 2-formyl-5,10,15,20-tetraphenylporphyrin
CAS:112163-33-4
CAS:112-84-5
CAS:43120-28-1
CAS:335-10-4