- +86-0533-2185556
- WhatsApp: +86 15965530500
- admin@hangyubiotech.com

pd_meltingpoint:136.0 to 140.0 °C
Purity:99%
|
Source |
Nobiletin is a nonpolar polymethoxylated flavone (PMFs) found in the citrus peel of various fruits, such as tangerine. Its chemical structure is similar to tangeretin, containing the typical flavonoid structure and six methoxyl groups (one more than tangeretin). |
|
structure and hydrogen bonding |
The crystal structure study on NOB shows that the chromene and the arene rings are almost in the same plane. The C atoms of the two methoxy groups in the arene ring are also in the same plane, while the C atoms of four methoxy groups linking to the chromene ring are not in parallel. This conformational characteristic of chiral structure is indicated by the covalent bond rotation between the arene and the chromene rings and the conformational alternations of methoxy groups. |
|
benefits |
Nobiletin improved cognitive deficits and the pathological features of AD, such as Aβ pathology, hyperphosphorylation of tau, and oxidative stress, in animal models of AD. In addition, nobiletin improved motor and cognitive deficits in PD animal models. These observations suggest that nobiletin has the potential to become a novel drug for the treatment and prevention of neurodegenerative diseases such as AD and PD. |
|
Application |
Nobiletin has been shown to exhibit antioxidant, anti-inflammatory, antitumor/cancer, and antiangiogenic activity in vitro and in vivo. Animal and in vitro data also demonstrate nobiletin's potential ability to suppress bone loss, lower cholesterol and atherosclerosis, and improve hyperglycemia and insulin resistance. |
|
Definition |
ChEBI: A methoxyflavone that is flavone substituted by methoxy groups at positions 5, 6, 7, 8, 3' and 4' respectively. |
InChI:InChI=1/C21H22O8/c1-23-13-8-7-11(9-15(13)24-2)14-10-12(22)16-17(25-3)19(26-4)21(28-6)20(27-5)18(16)29-14/h7-10H,1-6H3
[Figure not available: see fulltext.] A ...
The present invention provides a process...
We present a practical synthesis of the ...
Abstract: A series of polymethoxyflavono...
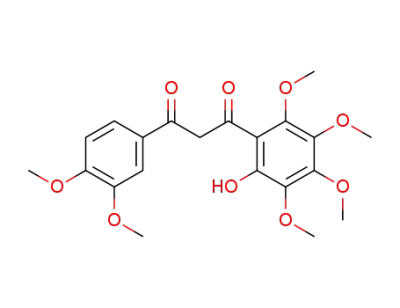
1-(2-hydroxy-3,4,5,6-tetramethoxyphenyl)-3-(3,4-dimethoxyphenyl)-1,3-propanedione

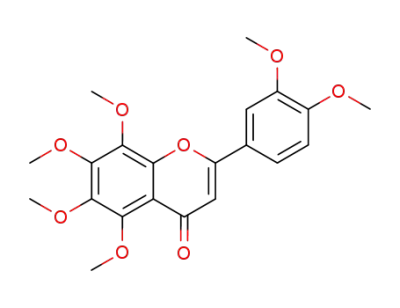
nobiletin
| Conditions | Yield |
|---|---|
|
With
trifluoroacetic acid;
In
methanol;
at 20 - 50 ℃;
for 24h;
|
98% |
|
With
sulfuric acid;
|
|
|
With
sulfuric acid;
In
acetic acid;
for 0.05h;
microwave irradiation;
|
2.03 g |
|
With
trifluoroacetic acid;
In
methanol;
at 50 ℃;
for 24h;
|
2.9 g |
|
With
trifluoroacetic acid;
In
methanol;
at 50 ℃;
for 24h;
Inert atmosphere;
|
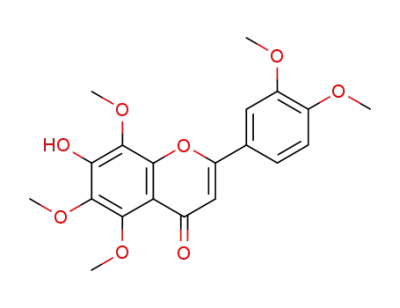
7-hydroxy-3’,4’,5,6,8-pentamethoxyflavone

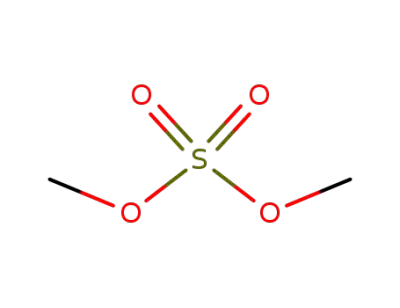
dimethyl sulfate


nobiletin
| Conditions | Yield |
|---|---|
|
With
potassium carbonate;
In
acetone;
at 65 ℃;
for 5h;
|
80% |
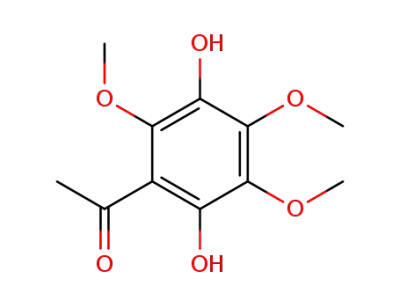
2',5'-dihydroxy-3',4',6'-trimethoxyacetophenone
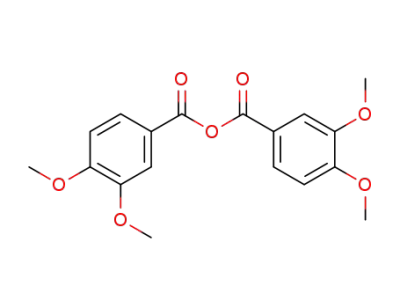
3,4-dimethoxybenzoic anhydride

1-(2-hydroxy-3,4,5,6-tetramethoxyphenyl)-3-(3,4-dimethoxyphenyl)-1,3-propanedione

dimethyl sulfate
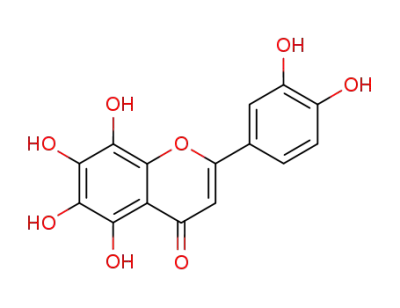
5-hydroxy-3',4',6,7,8-pentamethoxyflavone
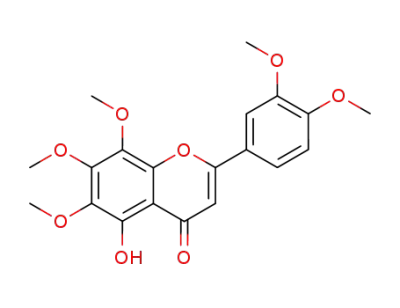
5-hydroxy-6,7,8,3',4'-pentamethoxyflavone
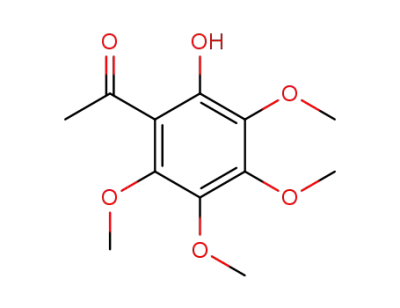
2-hydroxy-3,4,5,6-tetramethoxyacetophenone
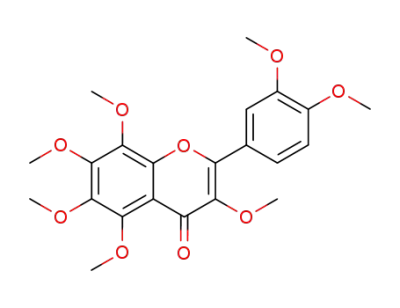
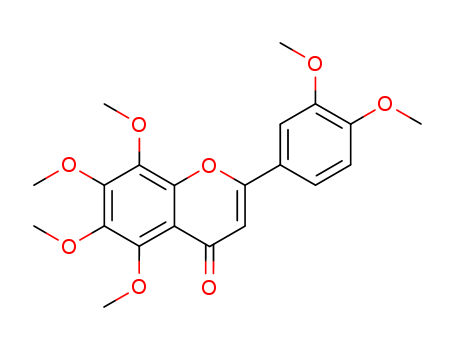
3,3',4',5,6,7,8-heptamethoxyflavone
CAS:112163-33-4
CAS:112-34-5
CAS:42464-96-0
CAS:53863-99-3