- +86-0533-2185556
- WhatsApp: +86 15965530500
- admin@hangyubiotech.com
Your Location:Home >Products >API >22112-79-4
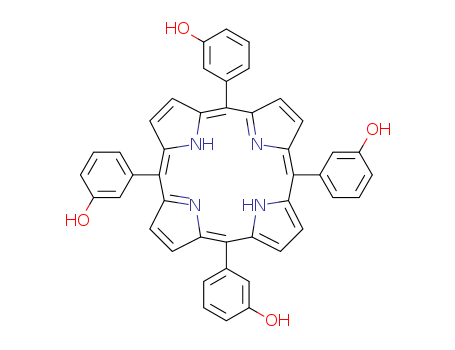

pd_meltingpoint:>320 °C
Purity:99%
The development of new antimicrobial str...
Abstract Several new derivatives of tetr...
Porphyrin compounds having groups that m...
Three p-hydroxy and m-hydroxy amphiphili...
Ideal reaction conditions were found to ...
Metalloporphyrins (and porphyrins) are w...
A new methodology for the synthesis of a...
The invention discloses a production met...
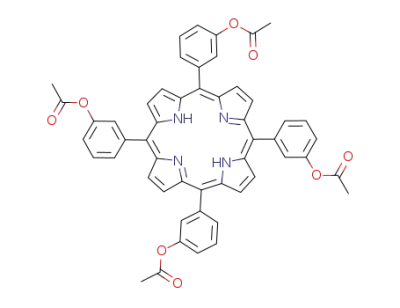
5,10,15,20-tetrakis(3-acetoxyphenyl)-porphyrin

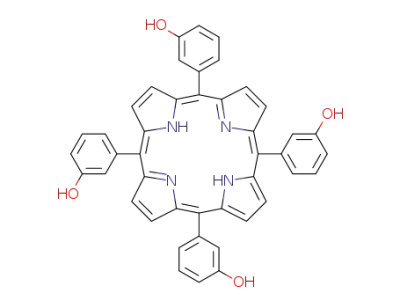
meso-5,10,15,20-tetra(3-hydroxyphenyl)porphine
| Conditions | Yield |
|---|---|
|
With
hydrogenchloride;
In
ethanol; water;
for 1h;
Reflux;
|
97% |
|
5,10,15,20-tetrakis(3-acetoxyphenyl)-porphyrin;
With
hydrogenchloride;
In
ethanol; water;
at 90 - 100 ℃;
for 1.5h;
With
sodium hydroxide;
at 20 ℃;
for 1h;
In
ethyl acetate;
at 20 ℃;
|
89% |
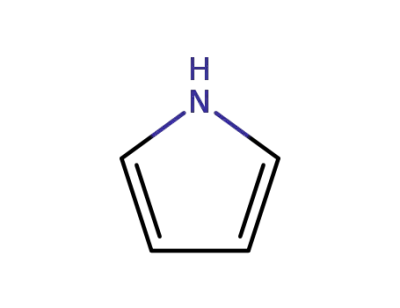
pyrrole

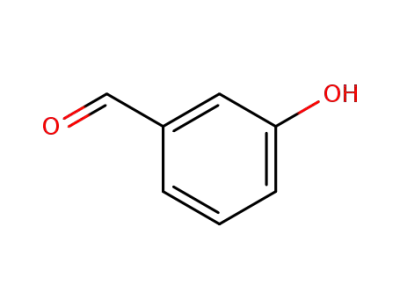
meta-hydroxybenzaldehyde


meso-5,10,15,20-tetra(3-hydroxyphenyl)porphine
| Conditions | Yield |
|---|---|
|
With
toluene-4-sulfonic acid;
In
N,N-dimethyl-formamide;
|
70% |
|
pyrrole; meta-hydroxybenzaldehyde;
With
propionic acid;
In
cyclohexane;
at 146.2 ℃;
for 0.4h;
under 3040.2 Torr;
In
cyclohexane;
for 0.9h;
Reflux;
|
49.2% |
|
pyrrole; meta-hydroxybenzaldehyde;
In
cyclohexane; propionic acid;
at 146.2 ℃;
for 0.4h;
under 3040.2 Torr;
Large scale;
Green chemistry;
With
air;
In
cyclohexane; propionic acid;
for 0.9h;
Large scale;
Green chemistry;
|
46.3% |
|
With
propionic acid; nitrobenzene;
at 200 ℃;
for 0.0833333h;
Microwave irradiation;
Sealed vessel;
|
36% |
|
In
water;
at 199.84 ℃;
for 0.166667h;
under 12001.2 Torr;
Microwave irradiation;
Green chemistry;
|
11% |
|
With
acetic acid;
In
nitrobenzene;
at 120 ℃;
for 1h;
Inert atmosphere;
|
10% |
|
With
acetic acid; nitrobenzene;
at 120 ℃;
for 1h;
|
10% |
|
In
propionic acid;
for 1.5h;
Reflux;
|
9% |
|
With
propionic acid;
at 130 ℃;
for 1h;
|
4.4% |
|
With
hydrogenchloride; sodium dodecyl-sulfate; 2,3-dicyano-5,6-dichloro-p-benzoquinone;
Product distribution;
multistep reaction; porphirin yield versus aldehyde concentration;
|
|
|
With
propionic acid;
at 20 - 140 ℃;
for 14h;
Heating / reflux;
|
|
|
With
trifluoromethane sulfonyl chloride;
In
dichloromethane;
at 20 ℃;
Inert atmosphere;
|
|
|
In
propionic acid;
for 14h;
Heating / reflux;
|
|
|
pyrrole; meta-hydroxybenzaldehyde;
With
propionic acid; propionic acid anhydride;
In
ethanol;
at 140 ℃;
for 1h;
In
ethanol;
at 20 ℃;
for 1h;
|
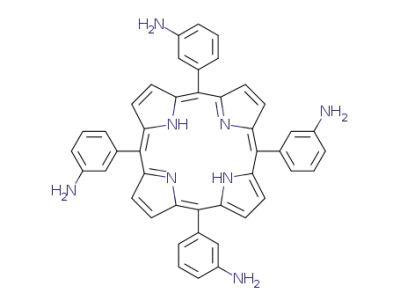
5,10,15,20-mesotetrakis(3-aminophenyl)-21H,23H-porphyrine

pyrrole

meta-hydroxybenzaldehyde
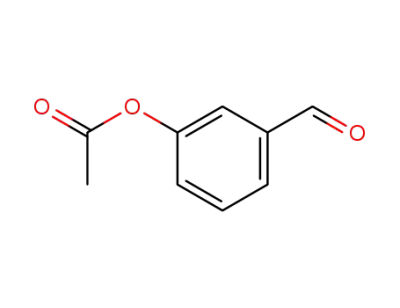
3-formylphenyl acetate
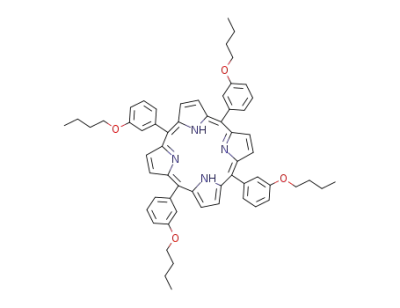
tetra(m-butoxyphenyl)porphin
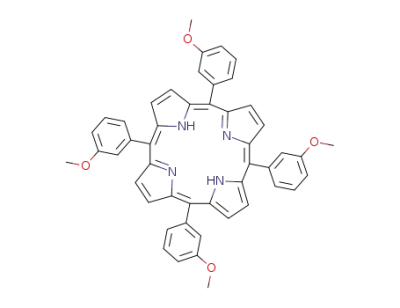
meso-tetra(m-methoxyphenyl)porphyrin
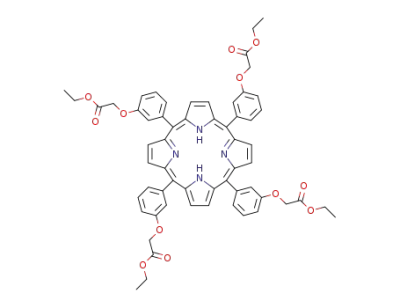
{3-[(1Z,4Z,9Z,15Z)-10,15,20-Tris-(3-ethoxycarbonylmethoxy-phenyl)-porphyrin-5-yl]-phenoxy}-acetic acid ethyl ester
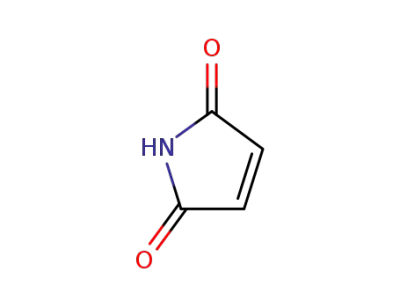
maleiimide
CAS:112163-33-4
CAS:112-84-5
CAS:53863-99-3
CAS:855-97-0