- +86-0533-2185556
- WhatsApp: +86 15965530500
- admin@hangyubiotech.com
Your Location:Home >Products >Customization >262-05-5
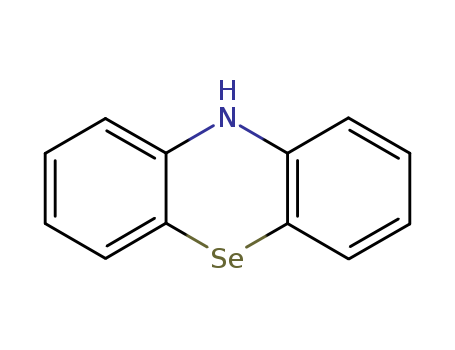

Purity:99%
|
Chemical Composition and Structure |
10H-Phenoselenazine is a chemical compound which consists of carbon, hydrogen, nitrogen, and selenium atoms. The compound exhibits specific chemical properties, including a melting point of 194-196°C (measured in benzene or hexane) and a predicted boiling point of 424.1±37.0°C. |
|
Production Methods |
10H-Phenoselenazine can be synthesized through a chemical reaction using (2-nitrobenzene)phenylselenyl ether as the raw material under nitrogen conditions in the presence of a reducing agent such as triphenylphosphine. |
|
General Description |
10H-Phenoselenazine is a chemical compound with the molecular formula C13H9NSe. It is a heterocyclic compound containing a selenazole ring, and it is also known as benzoselenadiazole. 10H-Phenoselenazine is a potential candidate for pharmaceutical research due to its diverse biological activities, including anti-cancer, anti-bacterial, and anti-inflammatory properties. The compound has shown promising results in inhibiting the growth of cancer cells and reducing inflammation in experimental studies. Its unique structure and potential pharmacological properties make 10H-Phenoselenazine an interesting target for further research and development in the field of medicinal chemistry. |
InChI:InChI=1/C12H9NSe/c1-3-7-11-9(5-1)13-10-6-2-4-8-12(10)14-11/h1-8,13H
A new twisted donor-acceptor-donor (D-A-...
Heavy-atom-modified chalcogen-fused tria...
The TeII/TeIII-catalyzed dehydrogenative...
Targeting energy metabolism in Mycobacte...
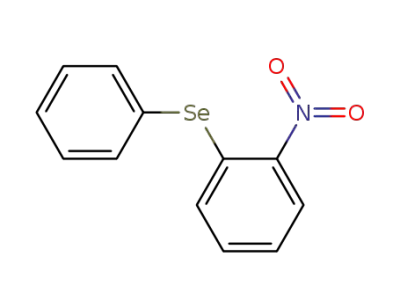
2-(phenylseleno)nitrobenzene

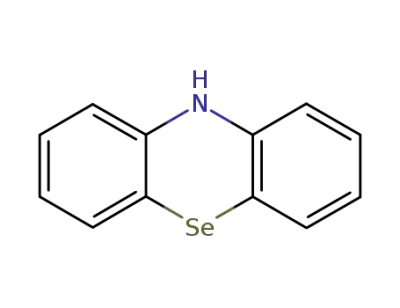
phenoselenazine
| Conditions | Yield |
|---|---|
|
With
triphenylphosphine;
In
1,2-dichloro-benzene;
at 180 ℃;
for 30h;
Reagent/catalyst;
Solvent;
Concentration;
Inert atmosphere;
|
92% |
|
With
triphenylphosphine;
In
1,2-dichloro-benzene;
at 180 ℃;
for 24h;
Inert atmosphere;
Sealed tube;
Schlenk technique;
|
68% |
|
With
triphenylphosphine;
In
1,2-dichloro-benzene;
at 180 ℃;
for 24h;
Inert atmosphere;
|
54% |
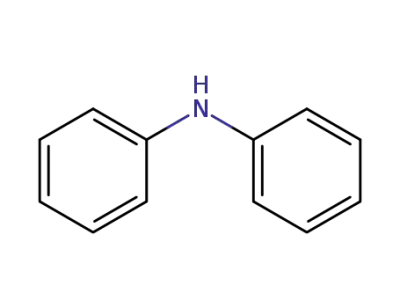
diphenylamine


phenoselenazine
| Conditions | Yield |
|---|---|
|
With
sulenium monochloride;
In
toluene;
for 5h;
Heating / reflux;
|
20% |
|
With
selenium(IV) oxide; selenium; iodine;
In
sulfolane;
at 150 ℃;
for 5h;
Sealed tube;
|
20% |
|
With
selenium(IV) oxide; selenium; iodine;
In
sulfolane;
at 150 ℃;
|
16% |
|
With
selenium tetrachloride; benzene;
|
|
|
With
chloroform; diselenium dichloride;
|
|
|
With
selenium(IV) oxide; selenium; iodine;
In
sulfolane;
|

diphenylamine

2-(phenylseleno)nitrobenzene
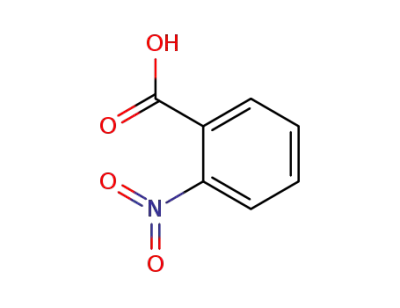
ortho-nitrobenzoic acid
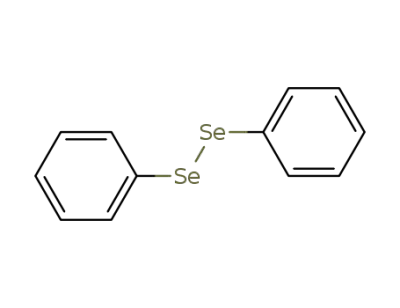
diphenyl diselenide
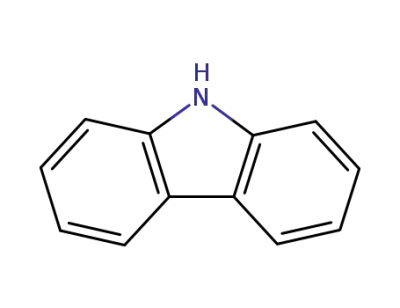
9H-carbazole
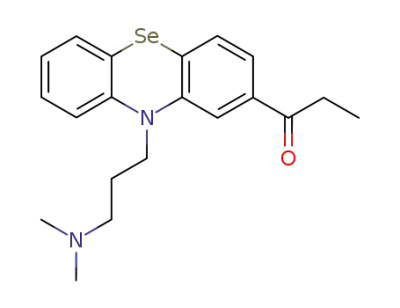
10-<3-Dimethylamino-propyl>-2-propionyl-phenselenazin
CAS:115473-15-9
CAS:1173-88-2
CAS:64-77-7
CAS:7499-19-6