- +86-0533-2185556
- WhatsApp: +86 15965530500
- admin@hangyubiotech.com
Your Location:Home >Products >Customization >3373-59-9
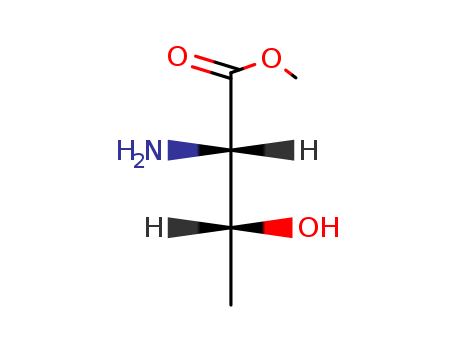

pd_meltingpoint:213-214 °C
Purity:99%
|
General Description |
L-Threonine methyl ester is a chemical compound that is derived from the amino acid L-threonine. It is a white crystalline substance that is commonly used in the synthesis of pharmaceuticals, agrochemicals, and other fine chemicals. L-threonine methyl ester is known for its role as a building block in the formation of various proteins and enzymes in biological systems. It is also used as a chiral reagent in organic synthesis and as a precursor in the production of various pharmaceutical compounds. Additionally, L-threonine methyl ester has potential applications in the fields of medicine, nutrition, and biotechnology. |
InChI:InChI=1/C5H11NO3/c1-3(7)4(6)5(8)9-2/h3-4,7H,6H2,1-2H3
Structurally diverse novel glycopeptoids...
Pyrazole, pyrazolone, and aminopyrazolon...
Th synthesis of Δ1-carbapenem and two β-...
The present invention relates to novel t...
A method for the selective tryptophan mo...
The invention provides a synthesis metho...
The present invention relates to novel t...
methanol

L-threonine

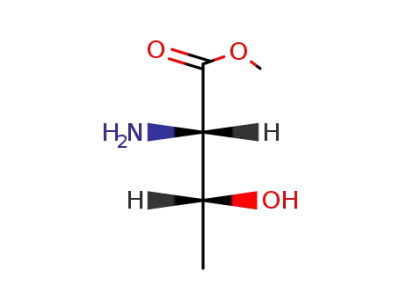
L-threonine methyl ester
| Conditions | Yield |
|---|---|
|
With
thionyl chloride;
at 0 ℃;
for 25h;
Temperature;
Reflux;
|
80% |
|
With
hydrogenchloride;
Heating;
|
|
|
With
hydrogenchloride;
for 5h;
Yield given;
Heating;
|
|
|
With
thionyl chloride;
for 12h;
Ambient temperature;
|
|
|
methanol; L-threonine;
With
hydrogenchloride;
With
ammonia;
In
diethyl ether;
|
|
|
With
thionyl chloride;
In
methanol;
at 0 - 23 ℃;
for 18h;
|
|
|
With
thionyl chloride;
at 0 - 25 ℃;
|
|
|
With
thionyl chloride;
for 2h;
Heating;
|
|
|
With
thionyl chloride;
at 20 ℃;
for 18h;
|
|
|
With
thionyl chloride;
|
|
|
With
chloro-trimethyl-silane;
at 80 ℃;
for 8h;
|
|
|
With
thionyl chloride;
at 0 - 20 ℃;
for 24h;
|
|
|
With
thionyl chloride;
at 0 - 75 ℃;
for 6h;
|
|
|
With
acetyl chloride;
at 0 ℃;
for 20h;
Reflux;
|
|
|
With
thionyl chloride;
at 3 - 45 ℃;
Large scale;
|
|
|
With
thionyl chloride;
at 0 ℃;
Reflux;
|
|
|
With
thionyl chloride;
at 0 - 25 ℃;
for 18h;
Industrial scale;
|
|
|
With
thionyl chloride;
Cooling with ice;
|
|
|
With
hydrogenchloride;
for 0.5h;
Reflux;
|
|
|
With
thionyl chloride;
Reflux;
|
|
|
With
thionyl chloride;
at 20 - 25 ℃;
Large scale;
|
|
|
With
thionyl chloride;
at 10 - 45 ℃;
|
|
|
With
thionyl chloride;
at 0 ℃;
|
|
|
With
thionyl chloride;
at -5 ℃;
Temperature;
Reflux;
|
|
|
With
thionyl chloride;
at 0 ℃;
for 3h;
Reflux;
|
|
|
With
thionyl chloride;
at 0 ℃;
for 3h;
Reflux;
|
8 g |
|
methanol;
With
thionyl chloride;
at 0 ℃;
for 0.166667h;
L-threonine;
for 2h;
Reflux;
|
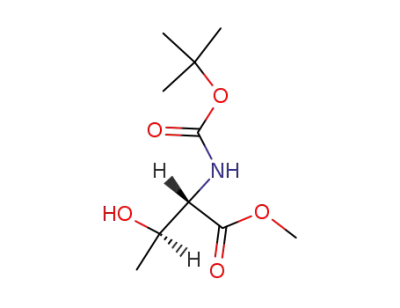
N-(tert-butoxycarbonyl)-l-threonine methyl ester


L-threonine methyl ester
| Conditions | Yield |
|---|---|
|
With
toluene-4-sulfonic acid;
In
acetone;
at 10 - 25 ℃;
for 2h;
|
70% |

methanol
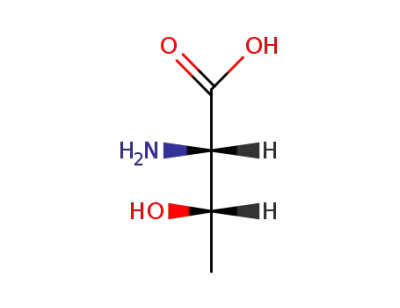
(2S,3S)-2-amino-3-hydroxybutanoic acid
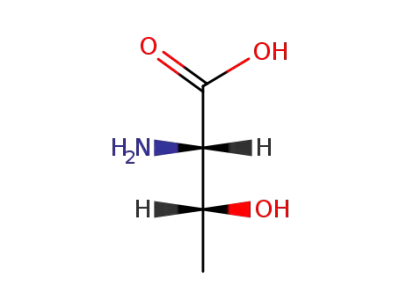
DL-threonine
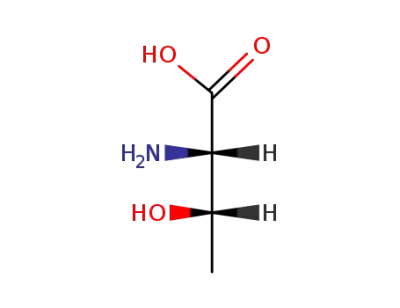
rac-allo-threonine
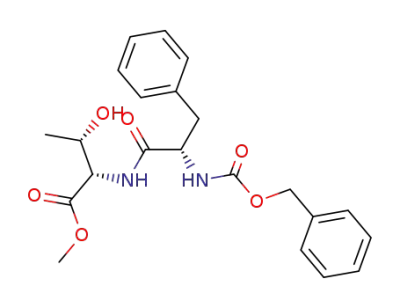
(2S,3S)-2-((S)-2-Benzyloxycarbonylamino-3-phenyl-propionylamino)-3-hydroxy-butyric acid methyl ester
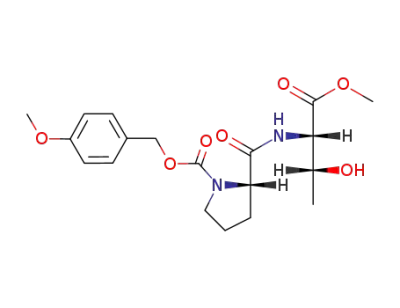
Z(OMe)-Pro-Thr-OMe
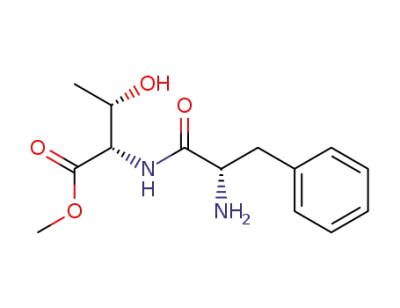
(2S,3S)-2-((S)-2-Amino-3-phenyl-propionylamino)-3-hydroxy-butyric acid methyl ester
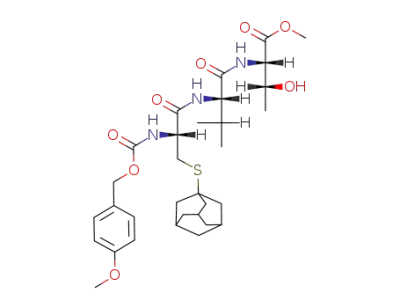
Z(OMe)-Cys(Ad)-Val-Thr-OMe
CAS:112163-33-4
CAS:112-84-5
CAS:96081-16-2
CAS:22112-84-1