- +86-0533-2185556
- WhatsApp: +86 15965530500
- admin@hangyubiotech.com
Your Location:Home >Products >API >22112-84-1
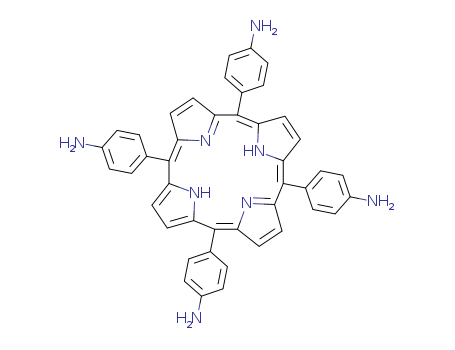

Purity:99%
InChI:InChI=1/C44H34N8/c45-29-9-1-25(2-10-29)41-33-17-19-35(49-33)42(26-3-11-30(46)12-4-26)37-21-23-39(51-37)44(28-7-15-32(48)16-8-28)40-24-22-38(52-40)43(36-20-18-34(41)50-36)27-5-13-31(47)14-6-27/h1-24,49-50H,45-48H2/b41-33-,41-34-,42-35-,42-37-,43-36-,43-38-,44-39-,44-40-
In order to reduce the toxicity and side...
We report an in-depth study on meso-aryl...
Next-generation photodynamic therapy age...
A novel 5,10,15,20-tetrakis[4-(3,5-dioct...
Presented here is a method for rapidly t...
Condensation of p-nitrobenzaldehyde with...
Recently, nitrogen-doped porous carbon s...
In this work, we firstly prepared new se...
Synthesis and processing of two-or three...
Three new porphyrin derivatives were syn...
Two three-dimensional (3D) porphyrin cov...
Fluorescence-switch-based logic devices ...
The chemical fixation of carbon dioxide ...
Developing large-scale electrocatalysts ...
Abstract In this work, two Schiff base p...
The emergence of drug-resistant pathogen...
A novel one-dimensional covalent organic...
Cobalt(II) meso-tetrakis(4-hexadecylamid...
Due to the existing challenges in the sy...
Dye-sensitized solar cells (DSSCs) were ...
In this work, a series of novel meso-tet...
In this work, 5, 10, 15, 20-Tetra-(4-ami...
Novel nanostructure, pH-thermo dual resp...
Three novel porphyrin-Schiff base conjug...
Aggregation of two porphyrin derivatives...
A cationic manganese porphyrin binds str...
An improved synthetic procedure is devel...
A series of porphyrin-containing polyimi...
Poly-5,10,15,20-tetrakis(p-aminophenyl)p...
The targeted synthesis of an efficient, ...
pyrrole

4-aminobenzaldehyde

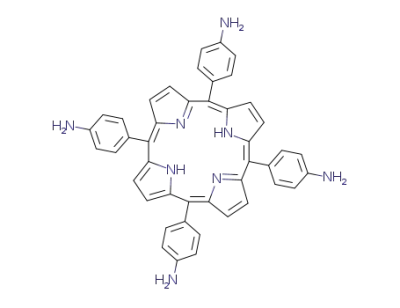
tetrakis(4-aminophenyl)porphyrin
| Conditions | Yield |
|---|---|
|
With
toluene-4-sulfonic acid;
In
N,N-dimethyl-formamide;
|
74% |
|
With
butyric acid;
In
N,N-dimethyl-formamide;
at 60 - 135 ℃;
|
55% |

pyrrole

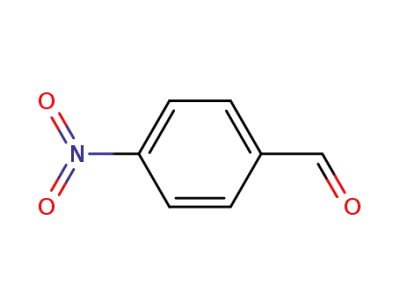
4-nitrobenzaldehdye


tetrakis(4-aminophenyl)porphyrin
| Conditions | Yield |
|---|---|
|
pyrrole; 4-nitrobenzaldehdye;
With
acetic anhydride;
In
propionic acid;
for 4h;
Reflux;
Inert atmosphere;
With
pyridine;
for 1.5h;
Reflux;
With
hydrogenchloride; tin(ll) chloride;
at 20 - 80 ℃;
for 3h;
|
37% |
|
4-nitrobenzaldehdye;
With
acetic anhydride; propionic acid;
at 80 ℃;
pyrrole;
at 135 ℃;
for 0.5h;
Further stages;
|
15.1% |
|
pyrrole; 4-nitrobenzaldehdye;
With
acetic anhydride;
In
acetic acid;
With
hydrogenchloride; tin(ll) chloride;
|
|
|
pyrrole; 4-nitrobenzaldehdye;
With
hydrogenchloride; acetic acid; tin(ll) chloride;
at 65 ℃;
|
|
|
pyrrole; 4-nitrobenzaldehdye;
With
acetic anhydride; propionic acid;
for 3h;
Reflux;
With
hydrogenchloride; tin(II) chloride dihdyrate;
|
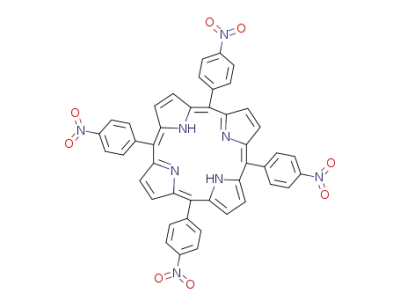
5,10,15,20-tetrakis(4-nitrophenyl)porphyrin

pyrrole

4-nitrobenzaldehdye
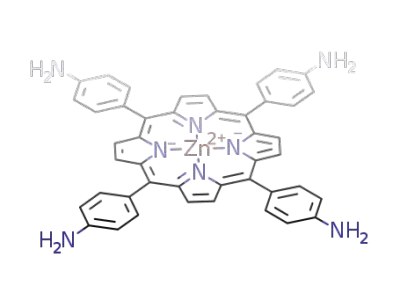
zinc(II) 5,10,15,20-tetrakis(4-aminophenyl)-21H,23H-porphine
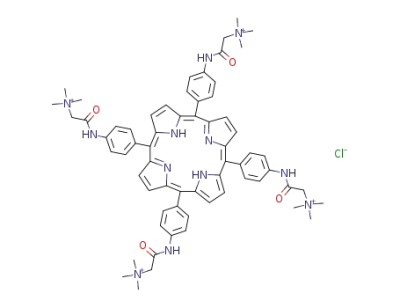
tetrakis(4-bentaineamidophenyl)porphyrin
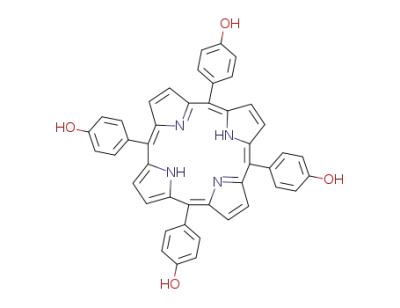
5,10,15,20-tetrakis(4'-hydroxyphenyl)porphyrin
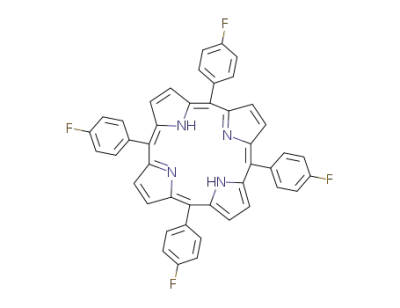
5,10,15,20-tetrakis(p-fluorophenyl)porphyrin
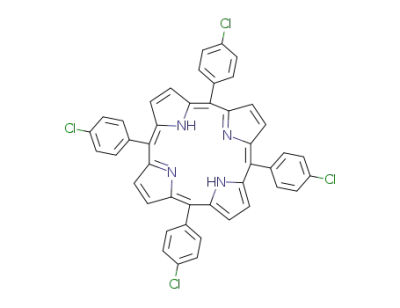
5,10,15,20-tetra-(4-chlorophenyl)porphyrin
CAS:112163-33-4
CAS:112-84-5
CAS:3373-59-9
CAS:129497-78-5