- +86-0533-2185556
- WhatsApp: +86 15965530500
- admin@hangyubiotech.com
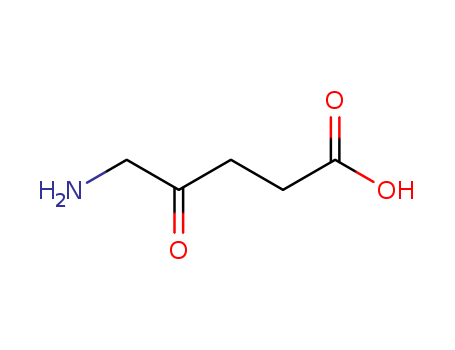

pd_meltingpoint:118-119 °C
Appearance:White crystalline powder
Purity:99%
|
Indications |
Aminolevulinic acid (ALA HCl, Levulan Kerastick) is indicated for the treatment of nonhyperkeratotic actinic keratosis of the face and scalp. It has two components, an alcohol solution vehicle and ALA HCl as a dry solid. The two are mixed prior to application to the skin. When applied to human skin, ALA is metabolized to protoporphyrin, which accumulates and on exposure to visible light produces a photodynamic reaction that generates reactive oxygen species (ROS).The ROS produce cytotoxic effects that may explain therapeutic efficacy. Local burning and stinging of treated areas of skin due to photosensitization can occur. |
|
Manufacturing Process |
1) Oxidation Step 2.27 g (10.0 mmol) of N-furfurylphthalimide was charged into a three-necked glass flask equipped with an oxygen feed tube, a thermometer, and a reflux condenser, and dissolved in 100 ml of anhydrous pyridine. After the addition of 7.0 mg of Rose Bengal, oxygen gas was fed at a rate of 20 ml/min at 10°- 20°C under irradiation by light. A 27 W white fluorescent lamp was used as a light source and the radiation was performed from the outside of the flask. After 7 hours, the irradiation was terminated and the pyridine was evaporated under reduced pressure to obtain 2.47 g of a light brown, semi-crystalline product. 2) Reduction Step (Hydrogenation) 2.00 g of the semi-crystalline solid obtained in (1) was dissolved in 40 ml of methanol and stirred at 50°C in a hydrogen atmosphere under atmospheric pressure in the presence of 200 mg of 5% palladium-on-carbon catalyst. After five hours, the reaction was terminated and the mixture was allowed to cool to room temperature. The catalyst was removed by filtration and methanol was evaporated to obtain 2.11 g of white crystals. The crystals were identified to be 5-phthalimidolevulinic acid by NMR analysis. The yield was 97%. 3) Hydrolysis Step 100 ml of 6 N hydrochloric acid was added to 2.11 g of the white crystals (2), and the mixture was heated under reflux for 5 hours. After evaporating the hydrochloric acid under reduced pressure, a brown solid product was obtained and dissolved in ethanol. Acetone was added to the solution and the crystals produced were collected by filtration to obtain 0.689 g of 5-aminolevulinic acid hydrochloride. The yield based on Nfurfurylphthalimide was 51%. NMR spectrum data conformed to 5-aminolevulinic acid hydrochloride |
|
Therapeutic Function |
Photosensitizer |
|
Biological Activity |
5-Aminolevulinic acid (5-ALA) is a precursor in the biosynthesis of porphyrins, including heme. The conversion of 5-ALA to protoporphyrins within tissues produces a photosensitive target that produces reactive oxygen species upon exposure to light.1 In this way, it is used in photodynamic therapy for a range of dermatological conditions, cancers, and other diseases. Also, oral administration of 5-ALA leads to the preferential accumulation of the fluorescent molecule protoporphyrin IX within certain types of cancer cells. This allows fluorescence-based identification of tumor tissue for accurate resection of diseased tissue. |
|
Enzyme inhibitor |
This key metabolic precursor (FW = 131.13 g/mol; CAS 106-60-5; pKa values = 4.05 and 8.90 at 25°C; Symbol: ALA), also known as daminolevulinic acid, is essential for the biosynthesis of metal ion-binding tetrapyrrole ring systems (porphyrins, chlorophylls, and cobalamins). In non-photosynthetic eukaryotes (animals, insects, fungi, protozoa, and alphaproteobacteria), d-aminolevulinic acid is produced by the enzyme ALA synthase, using glycine and succinyl CoA as substrates. In plants, algae, bacteria, and archaea, it is produced from glutamyl-tRNA and glutamate-1-semialdehyde. 5-Aminolevulinic acid inhibits (R)-3-amino-2- methylpropionate:pyruvate aminotransferase. ALA Phototherapy: Protoporphyrin IX, the immediate heme precursor is a highly effective tissue photosensitizer that is synthesized in four steps from 5- aminolevulinic acid. ALA synthesis is regulated via a feedback inhibition and gene repression mechanism linked to the concentration of free heme. In certain cell and tissue types, addition of exogenous ALA bypasses these regulation mechanisms, inducing uptake and synthesis of photosensitizing concentrations of Protoporphyrin IX, or PpIX. Topical application of ALA to certain malignant and non-malignant skin lesions, for example, can induce a clinically useful degree of lesion-specific photosensitization (e.g., superficial basal cell carcinomas show high response rate (~79%) after a single phototherapy treatment). ALA also induces localized tissue-specific photosensitization, when injected intradermally. In this sense, ALA and its methyl ester (methyl aminolevulinate, or MAL; trade name: Metvix?) are prodrugs that increase the amounts of the active drug (PpIX). |
|
General Description |
5-Amino-4-oxopentanoic acid (also known as aminolevulinic acid or ALA) is a key intermediate in the biosynthesis of tetrapyrrole macrocycles, such as porphyrins, which are essential for bioenergetic processes. In prebiotic chemistry, ALA has been demonstrated to react with other acyclic compounds under moderate conditions to form uroporphyrinogen, bypassing the need for porphobilinogen (PBG) and suggesting a plausible abiotic pathway for the synthesis of these critical macrocycles. This highlights its potential role in the origin of life and the formation of biologically significant molecules. |
InChI:InChI=1/C5H9NO3/c6-3-4(7)1-2-5(8)9/h1-3,6H2,(H,8,9)
5-Aminolevulinate synthase (ALAS) and 8-...
The evaluation of the photorelease of 5-...
Heme is an essential cofactor and signal...
5-Aminolevulinate synthase (EC 2.3.1.37)...
5-Aminolevulinic acid (ALA) and the biom...
The invention discloses a process for pr...
The invention relates to the field of or...
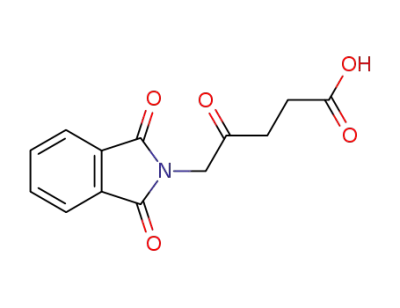
5-phthalimidyl levulinic acid

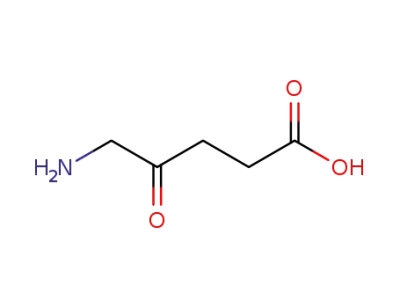
ALA
| Conditions | Yield |
|---|---|
|
With
hydrogenchloride;
In
water;
at 110 ℃;
for 12h;
|
81.7% |
|
With
hydrogenchloride;
|
C8H15NO3


ALA
| Conditions | Yield |
|---|---|
|
With
hydrogenchloride; cholesterol;
In
water;
at 70 ℃;
pH=7.5;
|
87% |
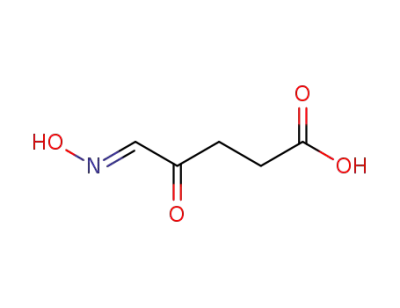
5-hydroxyimino-4-oxo-valeric acid

5-phthalimidyl levulinic acid
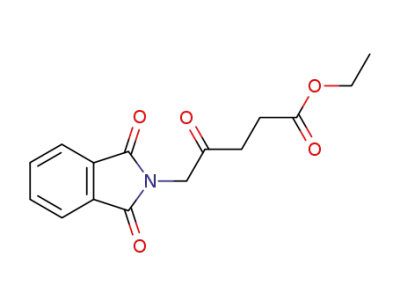
ethyl 5-(1,3-dioxoisoindolin-2-yl)-4-oxopentanoate
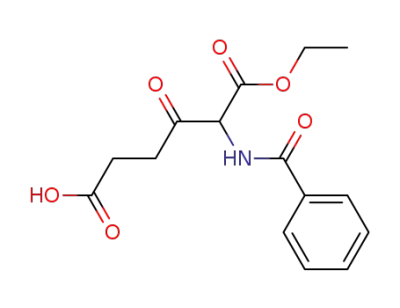
2-Benzoylamino-3-oxo-hexanedioic acid 1-ethyl ester
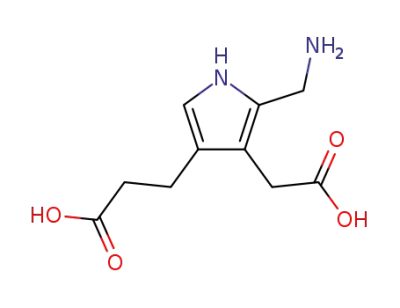
porphobilinogen
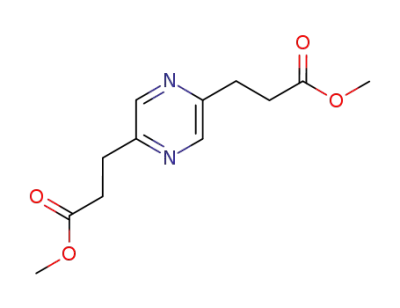
3-[5-(2-Methoxycarbonyl-ethyl)-pyrazin-2-yl]-propionic acid methyl ester
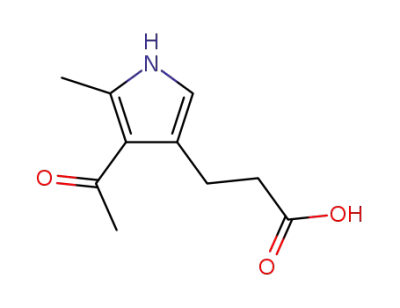
3-(4-acetyl-5-methyl-1H-pyrrol-3-yl)propanoic acid
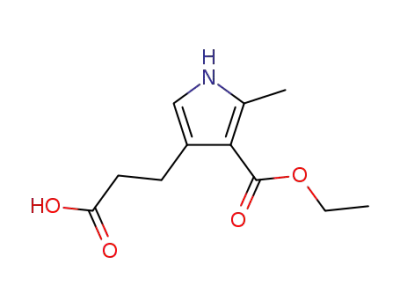
3-(2-carboxyethyl)-4-ethoxycarbonyl-5-methylpyrrole
CAS:112163-33-4
CAS:112-84-5
CAS:775304-57-9
CAS:13882-80-9