- +86-0533-2185556
- WhatsApp: +86 15965530500
- admin@hangyubiotech.com
Your Location:Home >Products >API >775304-57-9
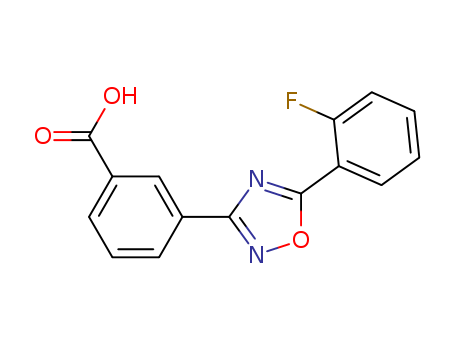

Purity:99%
|
Features |
Demonstrates oral bioavailability, and an appropriate safety toxicology profile. |
|
In vitro |
Compared with Gentamicin which is only active at much higher concentrations, PTC124 is a more potent nonsense-suppressing agent and exhibits 4-to 15-fold stimulation of read-through relative to controls. PTC124 (0.01-3 μM) promotes dose-dependent read-through of all three nonsense codons in HEK293 cells harboring LUC-190 nonsense alleles with the highest read-through at UGA, followed by UAG and then UAA, but it does not suppress multiple proximal nonsense codons. Like Gentamicin, PTC124 is most active when a pyrimidine (in particular cytosine, C) follows the nonsense codon. Consistent with the stable cell line reporter assay, PTC124 (17 μM) promotes significant production of dystrophin in primary muscle cells from Duchenne muscular dystrophy (DMD) patients or MDXMDX mice expressing dystrophin nonsense alleles. PTC124 selectively promotes ribosomal read-through of premature termination but not normal termination codons, even at concentrations substantially greater than the values achieving maximal activity. |
|
In vivo |
Due to functional recovery of dystrophin production, oral, intraperitoneal or combined dosing of PTC124 for 2-8 weeks partially rescues functional strength deficit in dystrophic muscles of MDX mice, and results in partial protection against contraction-induced injury in the extensor digitorum longus (EDL) muscles, as well as significant reductions in serum creatine kinase values. In Cftr-/-mice expressing a human CFTR-G542X transgene, subcutaneous or oral administration of PTC124 (~60 mg/kg) suppresses the G542X nonsense mutation in a dose-dependent manner, leading to a significant restoration of human (h)CFTR protein expression and function without any effect on nonsense-mediated mRNA decay (NMD) or other aspects of mRNA stability. PTC124 treatment (60 mg/kg) restores 29% of the normal intestinal transepithelial cAMP-stimulated shortcircuit currents observed in Cftr+/+ mice, displaying a significant advantage compared with Gentamicin. |
|
Synthesis |
The sequence to construct ataluren, which was described by the authors at PTC Therapeutics, commenced with commercially available methyl 3-cyanobenzoate (38). This ester was exposed to hydroxylamine in aqueous tert-butanol and warmed gently until the reaction was deemed complete. Then this mixture was treated with 2-fluorobenzoyl chloride dropwise and subsequently triethylamine dropwise. To minimize exotherm and undesired side products, careful control of the addition of reagents was achieved through slow dropwise addition of these liquid reagents. Upon complete consumption of starting materials and formation of amidooxime 39, the aqueous reaction mixture was then heated to 85 ℃ to facilitate 1,2,4-oxadiazole formation, resulting in the tricyclic ester 40 in excellent yield across the three steps. Finally, saponification of ester 40 through the use of sodium hydroxide followed by acidic quench gave ataluren (V) in 96% over the two-step sequence. |
InChI:InChI=1/C15H9FN2O3/c16-12-7-2-1-6-11(12)14-17-13(18-21-14)9-4-3-5-10(8-9)15(19)20/h1-8H,(H,19,20)
The invention discloses a preparation me...
The invention discloses a method for pre...
The present invention provides processes...
A direct C-5–H arylation of 3-substitute...
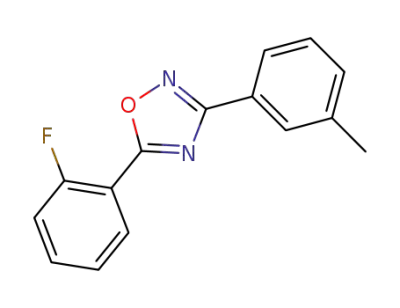
5-(2-fluorophenyl)-3-(3-methylphenyl)-1,2,4-oxadiazole

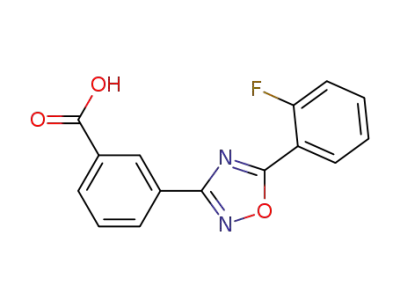
ataluren
| Conditions | Yield |
|---|---|
|
With
basic cobalt(II) carbonate; sodium bromide;
In
acetic acid;
at 95 ℃;
for 11h;
Green chemistry;
|
85% |
|
With
pyridine; potassium permanganate;
In
water;
at 85 ℃;
for 6.5h;
|
40% |
![3-[5-(2-fluorophenyl)-1,2,4-oxadiazol-3-yl]benzaldehyde](/upload/2026/5/e7bdff1f-c783-4063-9eec-ff0627aa875e.png)
3-[5-(2-fluorophenyl)-1,2,4-oxadiazol-3-yl]benzaldehyde


ataluren
| Conditions | Yield |
|---|---|
|
3-[5-(2-fluorophenyl)-1,2,4-oxadiazol-3-yl]benzaldehyde;
With
potassium dihydrogenphosphate; dihydrogen peroxide;
In
water; acetonitrile;
at 20 - 30 ℃;
With
sodium chlorite;
In
water; acetonitrile;
at 0 - 30 ℃;
for 5h;
|
90% |
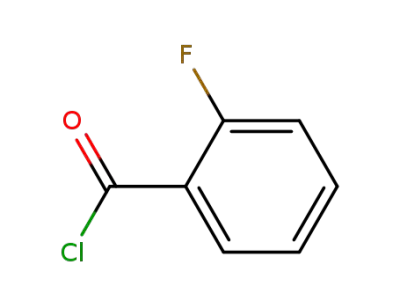
2-Fluorobenzoyl chloride
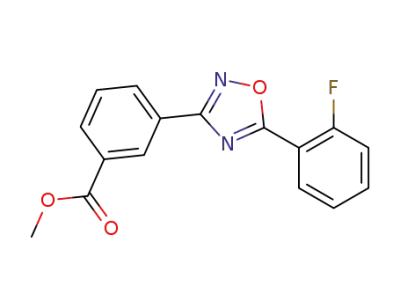
3-((5-(2-fluorophenyl))-(1,2,4-oxadiazol-3-yl))benzoic acid methyl ester
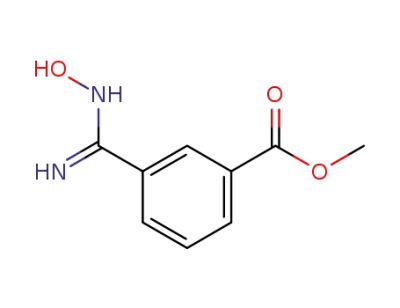
3-(N-hydroxycarbamimidoyl)-benzoic acid methyl ester

5-(2-fluorophenyl)-3-(3-methylphenyl)-1,2,4-oxadiazole

3-((5-(2-fluorophenyl))-(1,2,4-oxadiazol-3-yl))benzoic acid methyl ester
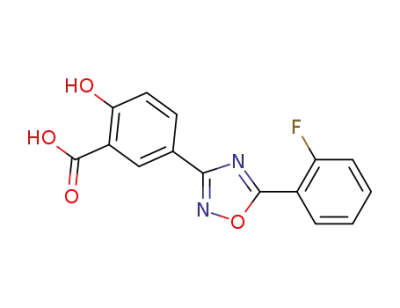
5-[5-(2-fluorophenyl)-[1,2,4]oxadiazol-3-yl]-2-hydroxybenzoic acid
CAS:117724-63-7
CAS:391210-10-9
CAS:2302-39-8
CAS:106-60-5