- +86-0533-2185556
- WhatsApp: +86 15965530500
- admin@hangyubiotech.com

pd_meltingpoint:245ºC (decomp)
Appearance:Crystals from water with 3 mol of water of crystallization.
Purity:99%
|
Characteristics |
L-leucovorin is used as an anti-tumor antidote and anti-megaloblastic anemia adjuvant. Its dosage is 1/2 of folinic acid. L-leucovorin does not need to be reduced by dihydrofolate reductase to participate in the use of folate as a The reaction is derived from carbon units, and L-leucovorin can actively or passively pass through the cell membrane; the basic function of L-leucovorin is the same as folic acid, but the effect is better than folic acid. At the same time, L-leucovorin also has the effect of stimulating the growth and maturation of white blood cells, which can improve the growth and maturation of white blood cells. |
|
Pharmacology |
Folic acid (pteroylglutamic acid), an essential water-soluble vitamin, consists of a pteridine ring joined to PABA (para-aminobenzoic acid) and glutamic acid.3?Folic acid is the most common of the many folate congeners that exist in nature and are essential for normal cellular metabolic functions. Folic acid is rarely called vitamin B9. After absorption, folic acid is reduced by dihydrofolic acid reductase (DHFR) to dihydrofolic acid and then tetrahydrofolic acid (THF), which accepts one-carbon groups. Tetrahydrofolic acid serves as the precursor for several biologically active forms of folic acid, including 5-formyltetrahydrofolic acid (5-formyl THF), which is best known as folinic acid, leucovorin, and citrovorum factor. |
|
Preparation |
In a 1L reaction flask, first add 0.45L of pure water, heat to 55-60℃ with stirring, then add 15g (0.0245mol, water 16.4%) of levofolinate, keep it at 55-60℃, and stir to It is almost dissolved, and the temperature is lowered to 0-10°C first, and an aqueous sodium carbonate solution (2.6g sodium carbonate (0.0245mol) dissolved in 45mL of pure water) is added dropwise, keeping the temperature at 0-10°C, after dripping, keep warm and stir for 1 hour. The precipitated solid is filtered out, the filter cake is washed once with 20 ml of pure water, and the filtrate is the sodium levofolinate solution. Put the filtrate into a 1L reaction flask, stir and lower the temperature to 0-5°C, control the temperature of the reaction system at 0-5°C, and add 1N dilute hydrochloric acid uniformly. When the pH of the acidification end point is 2.0-2.5, stop the dripping. Stir for about 30 minutes, then stand and age for 30 minutes, filter, and wash the filter cake twice with 75 mL of pure water. The wet product is freeze-dried to obtain 9.1 g of Folinic acid. Yield: 78% |
|
Definition |
ChEBI: A formyltetrahydrofolic acid in which the formyl group is located at position 5. |
InChI:InChI=1/C20H23N7O7/c21-20-25-16-15(18(32)26-20)27(9-28)12(8-23-16)7-22-11-3-1-10(2-4-11)17(31)24-13(19(33)34)5-6-14(29)30/h1-4,9,12-13,22H,5-8H2,(H,24,31)(H,29,30)(H,33,34)(H4,21,23,25,26,32)/t12-,13-/m0/s1
The one-carbon carrier of the formate ox...
A novel pharmaceutical composition for t...
Monoclonal antibodies that bind specific...
The inventive method for the production ...
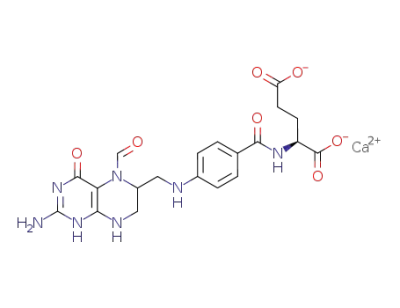
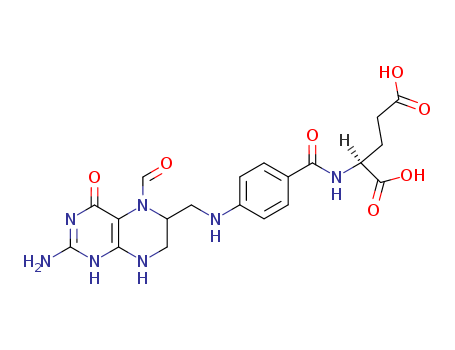
Leucovorin Calcium

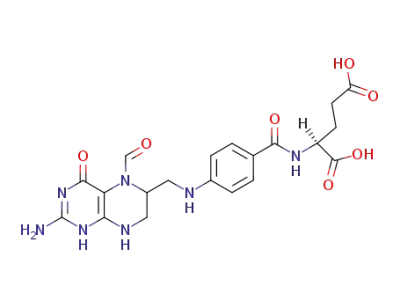
folinic acid
| Conditions | Yield |
|---|---|
|
With
hydrogenchloride;
In
water;
at 6 - 10 ℃;
for 4h;
pH=2.8 - 3.2;
|
99.4 % ee |
|
With
acetic acid;
In
water;
at 2 - 12 ℃;
pH=2.5 - 3.5;
|


folinic acid
| Conditions | Yield |
|---|---|
|
|
3.0 g (65%) |
|
|
|
|
|
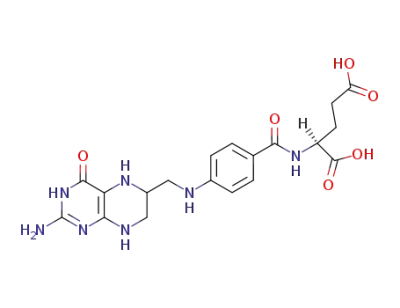
tetrahydrofolic acid
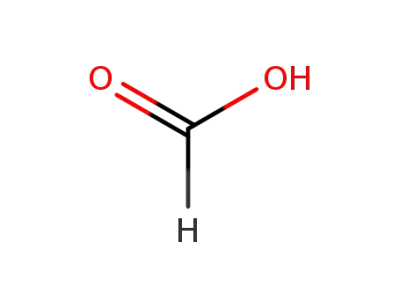
formic acid
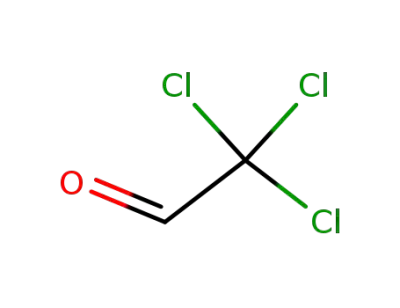
chloral
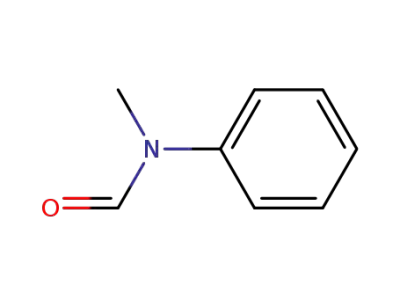
N-methyl-N-phenylformamide
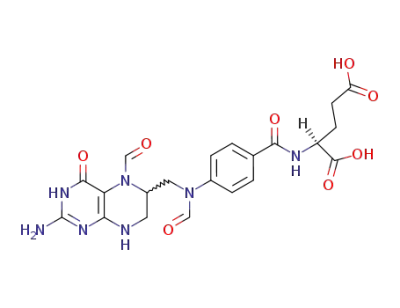
(6RS)-5,10-diformyl-5,6,7,8-tetrahydrofolic acid
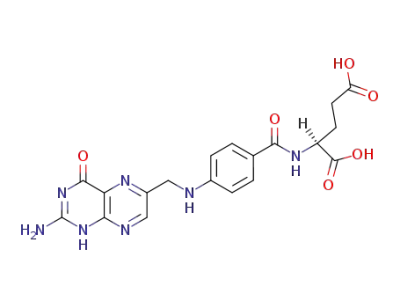
folate
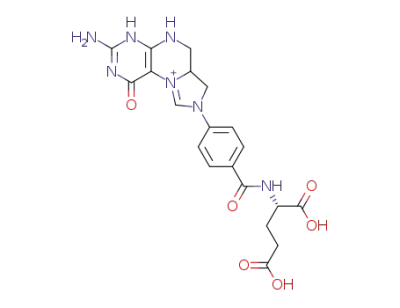
N5,N10-methenyl-tetrahydrofolic acid
CAS:112163-33-4
CAS:112-84-5
CAS:1218-35-5
CAS:24868-20-0