- +86-0533-2185556
- WhatsApp: +86 15965530500
- admin@hangyubiotech.com
Your Location:Home >Products >API >25122-46-7
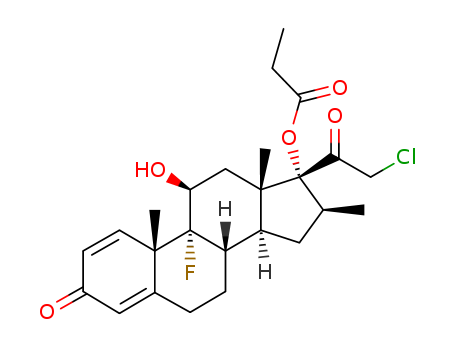

pd_meltingpoint:195.5-197 °C
Appearance:White crystalline powder
Purity:99%
|
Mechanism of Action |
Clobetasol Propionate is the propionate salt form of clobetasol, a topical synthetic corticosteroid with anti-inflammatory, anti-pruritic, and vasoconstrictive properties. Clobetasol propionate exerts its effect by binding to cytoplasmic glucocorticoid receptors and subsequently activates glucocorticoid receptor mediated gene expression. This results in the synthesis of certain anti-inflammatory proteins, while inhibiting the synthesis of certain inflammatory mediators. Specifically, clobetasol propionate appears to induce phospholipase A2 inhibitory proteins, thereby controlling the release of the inflammatory precursor arachidonic acid from membrane phospholipids by phospholipase A2. |
|
Indications |
Clobetasol propionate is the 17-O-propionate ester of clobetasol and it derives from a clobetasol and a propionic acid. A potent corticosteroid, it is used to treat various skin disorders, including exzema and psoriasis. Clobetasol propionate was patented in 1968 and came into medical use in 1978. It is available as a generic medication. Clobetasol propionate is used for the treatment of various skin disorders including eczema, herpes labialis, psoriasis, and lichen sclerosus. It is also used to treat several auto-immune diseases, including alopecia areata, lichen planus (auto immune skin nodules), and mycosis fungoides (T-cell skin lymphoma). It is used as first-line treatment for both acute and chronic GVHD of the skin. |
|
Definition |
ChEBI: The 17-O-propionate ester of clobetasol. A potent corticosteroid, it is used to treat various skin disorders, including exzema and psoriasis. |
InChI:InChI=1/C25H32ClFO5/c1-5-21(31)32-25(20(30)13-26)14(2)10-18-17-7-6-15-11-16(28)8-9-22(15,3)24(17,27)19(29)12-23(18,25)4/h8-9,11,14,17-19,29H,5-7,10,12-13H2,1-4H3/t14-,17?,18?,19-,22-,23-,24-,25-/m0/s1
The invention provides a clobetasol prop...
The invention relates to a method for th...
The invention provides a brand new synth...
The present invention relates to a use o...
C25H31ClO5

clobetasol-17-propionate
| Conditions | Yield |
|---|---|
|
With
hydrogen fluoride;
In
tetrahydrofuran; water;
at -5 - 0 ℃;
Solvent;
|
94.3% |
|
With
hydrogen fluoride;
In
chloroform; water;
at -40 ℃;
|
84% |
C25H31ClO5


clobetasol-17-propionate
| Conditions | Yield |
|---|---|
|
With
hydrogen fluoride; potassium hydroxide;
In
water; acetone;
at -40 - -30 ℃;
for 6h;
Temperature;
|
95% |
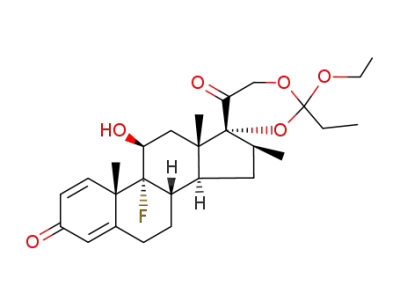
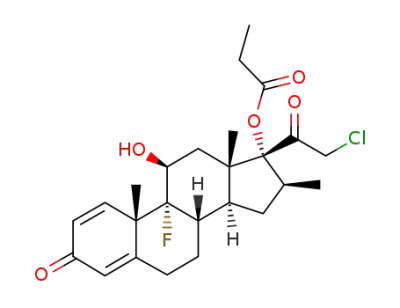
Betamethasone 17α,21-ethyl orthopropanoate
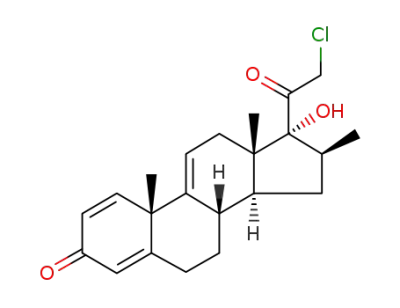
21-Chloro-17α-hydroxy-16β-methylpregna-1,4,9(11)-triene-3,20-dione
CAS:137504-86-0
CAS:98319-26-7
CAS:61748-93-4