- +86-0533-2185556
- WhatsApp: +86 15965530500
- admin@hangyubiotech.com
Your Location:Home >Products >API >191732-72-6
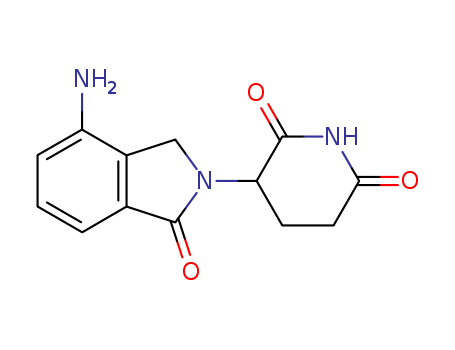

pd_meltingpoint:269-271 °C
Appearance:yellow solid
Purity:99%
Quality products make an important contribution to long-term revenue and profitability. Excellent chemical plant bulk supply Lenalidomide 191732-72-6
Lenalidomide is a thalidomide analog known to have immunomodulatory properties. Lenalidomide inhibits TNF-alpha production, stimulates T cells, reduces serum levels of the cytokines vascular endothelial growth factor (VEGF) and basic fibroblast growth factor (bFGF), and inhibits angiogenesis. This agent also promotes G1 cell cycle arrest and apoptosis of malignant cells. It is used in the therapy of multiple myeloma.
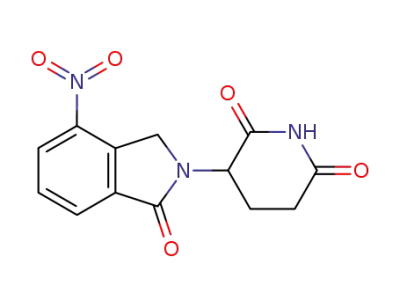
3-(4-nitro-1-oxoisoindolin-2-yl)piperidine-2,6-dione

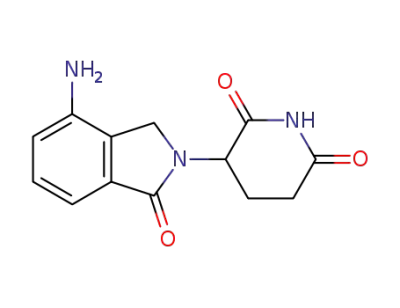
Lenalidomide
| Conditions | Yield |
|---|---|
|
With
5%-palladium/activated carbon; hydrogen;
In
ethanol;
at 40 ℃;
under 3000.3 Torr;
Pressure;
Temperature;
|
98.2% |
|
With
sodium sulfide; ethanol;
at 20 ℃;
for 1h;
|
98% |
|
With
sodium tetrahydroborate; ammonium chloride;
In
ethanol; water;
at 90 ℃;
for 1h;
Reagent/catalyst;
|
98% |
|
With
palladium 10% on activated carbon; hydrogen;
In
ethanol;
at 140 ℃;
for 0.00944444h;
under 11251.1 Torr;
Pressure;
Solvent;
Temperature;
Green chemistry;
|
96.11% |
|
With
palladium 10% on activated carbon; hydrogen;
In
methanol;
at 30 - 35 ℃;
for 4h;
under 1500.15 - 2250.23 Torr;
Reagent/catalyst;
Solvent;
Temperature;
Autoclave;
|
96% |
|
With
sodium hypophosphite; phosphoric acid; palladium 10% on activated carbon;
In
water;
at 60 ℃;
for 2h;
Reagent/catalyst;
Temperature;
Solvent;
|
95.95% |
|
With
palladium 10% on activated carbon; ammonium formate;
In
dimethyl sulfoxide;
at 30 - 40 ℃;
Temperature;
Solvent;
|
93.4% |
|
With
5%-palladium/activated carbon; hydrogen;
In
ethanol;
for 1h;
under 3040.2 Torr;
Reagent/catalyst;
|
92% |
|
With
palladium on activated charcoal; hydrogen;
In
ethanol;
|
92.8% |
|
With
iron; ammonium chloride;
In
ethanol; water;
at 90 ℃;
for 1.5h;
Reflux;
|
90% |
|
With
iron; acetic acid;
In
ethanol; water;
at 65 - 75 ℃;
Reagent/catalyst;
Solvent;
Temperature;
Inert atmosphere;
|
90.3% |
|
With
ammonium formate;
5% Pd(II)/C(eggshell);
In
methanol;
at 20 ℃;
for 2h;
|
89% |
|
With
tin(ll) chloride;
In
ethanol;
at 15 - 80 ℃;
for 2h;
|
85.6% |
|
With
iron; ammonium chloride;
In
ethanol; water;
at 60 - 80 ℃;
for 4h;
|
84% |
|
With
acetic acid; zinc;
In
methanol; N,N-dimethyl-formamide;
at 65 ℃;
for 0.25h;
Temperature;
Concentration;
Inert atmosphere;
|
84% |
|
With
palladium 10% on activated carbon; hydrogen;
In
methanol;
at 25 ℃;
for 10h;
under 9000.9 Torr;
Reagent/catalyst;
|
83.6% |
|
With
palladium 10% on activated carbon; ammonium formate;
In
N,N-dimethyl acetamide;
at 0 ℃;
for 14h;
Solvent;
|
83.5% |
|
With
palladium 10% on activated carbon; hydrogen;
In
water; N,N-dimethyl-formamide;
at 80 ℃;
under 2250.23 - 6000.6 Torr;
Green chemistry;
|
80.4% |
|
With
palladium 10% on activated carbon; hydrogen;
at 30 - 40 ℃;
|
80.1% |
|
With
palladium 10% on activated carbon; hydrogen;
In
methanol;
at 20 ℃;
for 2h;
Inert atmosphere;
|
80% |
|
3-(4-nitro-1-oxoisoindolin-2-yl)piperidine-2,6-dione;
With
hydrogen;
palladium 10% on activated carbon;
In
methanol; acetonitrile;
at 30 - 35 ℃;
Inert atmosphere;
With
hydrogenchloride;
In
isopropyl alcohol;
With
triethylamine;
In
methanol;
|
78% |
|
With
palladium on activated charcoal; hydrogen;
In
methanol; N,N-dimethyl-formamide;
at 20 ℃;
|
77.6% |
|
With
hydrogen;
palladium 10% on activated carbon;
In
N,N-dimethyl-formamide;
at 25 - 45 ℃;
under 2206.72 - 2942.29 Torr;
Product distribution / selectivity;
Parr shaker hydrogenator;
|
75.8% |
|
With
hydrogen;
palladium 10% on activated carbon;
In
methanol; N,N-dimethyl-formamide;
at 25 - 30 ℃;
under 2585.81 - 3102.97 Torr;
Product distribution / selectivity;
Parr shaker hydrogenator;
|
75.8% |
|
With
iron; ammonium chloride;
In
ethanol; water;
at 60 - 80 ℃;
for 4h;
Reagent/catalyst;
Solvent;
Temperature;
Green chemistry;
|
75% |
|
With
hydrogen;
palladium 10% on activated carbon;
In
methanol;
for 42h;
under 2362.74 Torr;
|
29% |
|
With
hydrogen;
palladium on activated charcoal;
In
methanol;
|
|
|
With
hydrogen;
palladium on activated charcoal;
|
|
|
With
hydrogen;
|
|
|
3-(4-nitro-1-oxoisoindolin-2-yl)piperidine-2,6-dione;
With
methanesulfonic acid; hydrogen;
palladium 10% on activated carbon;
In
water;
at 30 ℃;
for 3h;
under 4654.46 Torr;
With
sodium hydrogencarbonate;
In
water;
Product distribution / selectivity;
|
|
|
3-(4-nitro-1-oxoisoindolin-2-yl)piperidine-2,6-dione;
With
hydrogen;
palladium 10% on activated carbon;
In
methanol; acetonitrile;
at 30 - 35 ℃;
for 3.5h;
With
triethylamine;
In
methanol;
at 5 - 10 ℃;
|
|
|
With
hydrogen;
palladium 10% on activated carbon;
In
N,N-dimethyl-formamide;
for 3h;
under 2585.81 - 3102.97 Torr;
Product distribution / selectivity;
|
|
|
Multi-step reaction with 2 steps
1: hydrogen / palladium 10% on activated carbon / methanol / 18 h / 21 - 35 °C / 2400.24 Torr
2: sodium hydrogencarbonate / water / 65 - 70 °C / pH 8
With
hydrogen; sodium hydrogencarbonate;
palladium 10% on activated carbon;
In
methanol; water;
|
|
|
With
hydrogen;
palladium 10% on activated carbon;
In
N,N-dimethyl-formamide;
for 4h;
under 2585.81 - 3102.97 Torr;
|
|
|
With
hydrogen;
palladium 10% on activated carbon;
In
N,N-dimethyl-formamide;
at 60 - 65 ℃;
for 6h;
|
|
|
3-(4-nitro-1-oxoisoindolin-2-yl)piperidine-2,6-dione;
With
palladium on activated charcoal; hydrogen; acetic acid;
for 4h;
With
ammonia;
In
water;
at 10 - 15 ℃;
for 2h;
pH=7 - 7.5;
|
48 g |
|
With
palladium on activated charcoal; hydrogen;
In
methanol;
under 7600.51 Torr;
|
|
|
With
palladium on activated charcoal; hydrogen;
In
methanol;
at 20 ℃;
|
|
|
With
palladium 10% on activated carbon; hydrogen;
In
tetrahydrofuran;
|
|
|
With
palladium 10% on activated carbon; hydrogen;
In
methanol;
for 0.583333h;
under 2585.81 - 2844.39 Torr;
Time;
Autoclave;
|
6.9 g |
|
With
palladium on activated charcoal; hydrogen;
In
tetrahydrofuran; methanol;
at 20 ℃;
for 8h;
under 775.743 Torr;
|
|
|
With
palladium 10% on activated carbon; ammonia; hydrogen;
In
1-methyl-pyrrolidin-2-one;
for 12h;
under 3102.97 Torr;
Pressure;
Large scale;
|
|
|
With
palladium 10% on activated carbon; hydrogen;
In
N,N-dimethyl acetamide;
at 25 ℃;
under 4654.46 Torr;
Temperature;
Pressure;
|
480 g |
|
With
palladium 10% on activated carbon; hydrogen;
In
methanol;
at 20 ℃;
for 16h;
under 2068.65 Torr;
|
![2-[4-(N-benzyloxycarbonyl)amino-1,3-dihydro-1-oxo-2H-isoindol-2-yl]glutaramide](/upload/2026/5/243551b2-43b0-4c6d-8bfa-0afbedc9c20e.png)
2-[4-(N-benzyloxycarbonyl)amino-1,3-dihydro-1-oxo-2H-isoindol-2-yl]glutaramide


Lenalidomide
| Conditions | Yield |
|---|---|
|
2-[4-(N-benzyloxycarbonyl)amino-1,3-dihydro-1-oxo-2H-isoindol-2-yl]glutaramide;
In
N,N-dimethyl-formamide;
at 160 ℃;
With
N,N-dimethyl-formamide;
5% Pd(II)/C(eggshell);
In
methanol;
at 30 ℃;
for 2h;
|
72% |
The CAS number of Lenalidomide is 191732-72-6.
More information of Lenalidomide 191732-72-6 are:
|
CAS Number |
191732-72-6 |
|
Density |
1.46 g/cm3 |
|
Melting Point |
269-271 °C |
|
Boiling Point |
614 °C at 760 mmHg |
|
Flash Point |
325.1 °C |
|
Vapor Pressure |
5.2E-15mmHg at 25°C |
|
Refractive Index |
1.672 |
|
HS CODE |
29339900 |
|
PSA |
92.50000 |
|
LogP |
0.87770 |
|
Pka |
10.75±0.40(Predicted) |
Synonyms for Lenalidomide 191732-72-6:Lenalidomide(other anti-cancers);Revlimid (lenalidomide);Revlimid (lenalidomide)/191732-72-6;Lenalidomide ;3-(7-Amino-3-oxo-1H-isoindol-2-yl)piperidine-2,6-dione;Revlimid;
The chemical formula of Lenalidomide is C13H13N3O3 which containing 13 Carbon atoms,13 Hydrogen atoms,3 Nitrogen atoms and 3 Oxygen atoms,and the molecular weight of Lenalidomide is 259.265.
Lenalidomide is a kind of antitumor drugs that developed by American biological pharmaceutical companies. Its chemical structure is similar with thalidomide. It differing in the presence of an amino moiety in the 4-position and removal of one of the carbonyls of the phthaloyl ring. This derivative evolved from a structural-based effort to eliminate the adverse effects (somnolence, neuropathy, and teratogenicity) of thalidomide while maintaining or enhancing the appealing attributes. It has many functions such as anti-tumor, immune regulation and anti-angiogenesis. It can inhibit the secretion of inflammatory cytokines, and increase the secretion of peripheral blood mononuclear anti-inflammatory cytokines. Vitro tests show that lenalidomide can inhibit the hyperplasia of some cell lines such as namalwa cell. It can inhibit the growth of patients’ multiple myeloma cells and MM1S cell. In addition, lenalidomide also can inhibit the expression of oxidase-2 (COX-2), but it has no effect on COX-1. Two multicenter randomized double-blind placebo-controlled clinical studies evaluate the safety and curative effect of lenalidomide that is used for multiple myeloma. The primary efficacy end point of the studies is time to progression (TTP). The interim analysis shows that TTP of the combination group is significantly superior to dexamethasone group. Recent clinical research results show that lenalidomide not only has curative effect on treating MDS and MM, but also on treating myeloma, leukemia, metastatic renal cell carcinoma, solid tumor, idiopathic generalized amyloidosis and systemic bone marrow fibrosis disease with marrow unripe.
InChI:InChI=1/C13H13N3O3/c14-9-3-1-2-7-8(9)6-16(13(7)19)10-4-5-11(17)15-12(10)18/h1-3,10H,4-6,14H2,(H,15,17,18)
Relevant articles related to Lenalidomide:
|
Article |
Source |
|
Compound for targeted degradation of BTK protein |
- Paragraph 0458; 0468-0470, (2021/06/22) |
|
Method for preparing lenalidomide |
- Paragraph 0085-0090, (2020/06/09) |
Zibo Hangyu Biotechnology Development Co., Ltd is a quality supplier of Lenalidomide. Our main goal is customer satisfaction. Contact us to negotiate the best price for your business on Lenalidomide 191732-72-6.
CAS:112163-33-4
CAS:112-84-5
CAS:1369764-02-2
CAS:61049-69-2