- +86-0533-2185556
- WhatsApp: +86 15965530500
- admin@hangyubiotech.com
Your Location:Home >Products >API >249296-44-4

pd_meltingpoint:138.5 °C
Purity:99%
|
Synthesis |
Several modifications to the original synthesis have been reported in the literature, including an improved process scale synthesis of the last few steps.The Grignard reaction was initiated on a small scale by addition of 2-bromo fluorobenzene 113 to a slurry of Magnesium turnings and catalytic 1,2-dibromoethane in THF and heating the mixture until refluxing in maintained. To this refluxing mixture was added a mixture of the 2-bromo fluorobenzene 113 and cyclopentadiene 114 over a period of 1.5 h. After complete addition, the reaction was allowed to reflux for additional 1.5 h to give the Diels- Alder product 115 in 64% yield. Dihydroxylation of the olefin 115 by reacting with catalytic osmium tetraoxide in the presence of N-methylmorpholine N-oxide (NMO) in acetone: water mixture at room temperature provided the diol 116 in 89% yield. Oxidative cleavage of diol 116 with sodium periodate in biphasic mixture of water: DCE at 10oC provided di-aldehyde 117 which was immediately reacted with benzyl amine in the presence of sodium acetoxyborohydride to give benzyl amine 118 in 85.7% yield. The removal of the benzyl group was effected by hydrogenation of the HCl salt in 40-50 psi hydrogen pressure with 20% Pd(OH)2 in methanol to give amine hydrochloride 119 in 88% yield. Treatment of amine 119 with trifluoroacetic anhydride and pyridine in dichloromethane at 0oC gave trifluoroacetamide 120 in 94% yield. Dinitro compound 121 was prepared by addition of trifluoroacetamide 120 to a mixture of trifluoromethane sulfonic acid and nitric acid, which was premixed, in dichloromethane at 0oC. Reduction of the dinitro compound 121 by hydrogenation at 40-50 psi hydrogen in the presence of catalytic 5%Pd/C in isopropanol:water mixture provided the diamine intermediate 122 which was quickly reacted with glyoxal in water at room temperature for 18h to give compound 123 in 85% overall yield. The trifluoroacetamide 123 was then hydrolyzed with 2 M sodium hydroxide in toluene at 37-40oC for 2-3h followed by preparation of tartrate salt in methanol to furnish varenicline tartrate (XV). |
|
Drug interactions |
Potentially hazardous interactions with other drugs None known |
|
Metabolism |
Varenicline undergoes minimal metabolism with less than 10% excreted as metabolites. About 92% of a dose is excreted unchanged in the urine. Minor metabolites in urine include varenicline N-carbamoylglucuronide, N-glucosylvarenicline and hydroxyvarenicline. In circulation, varenicline comprises 91% of drug-related material. |
|
references |
[1] garrison gd, dugan se. varenicline: a first-line treatment option for smoking cessation. clin ther. 2009 mar;31(3):463-91. |
|
Definition |
ChEBI: An organic heterotetracyclic compound that acts as a partial agonist for nicotinic cholinergic receptors and is used (in the form of its tartate salt) as an aid to giving up smoking. |
|
Brand name |
Chantix (Pfizer). |
InChI:InChI=1/C13H13N3/c1-2-16-13-5-11-9-3-8(6-14-7-9)10(11)4-12(13)15-1/h1-2,4-5,8-9,14H,3,6-7H2
The present invention relates to a proce...
Provided is a cocrystal of varenicline a...
The present invention is directed to an ...
Provided herein is an impurity of vareni...
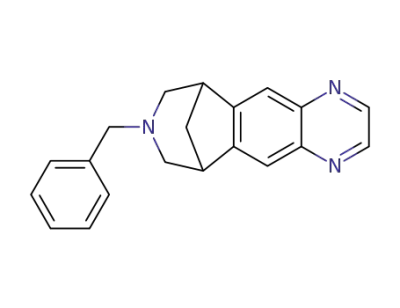
N-benzylvarenicline

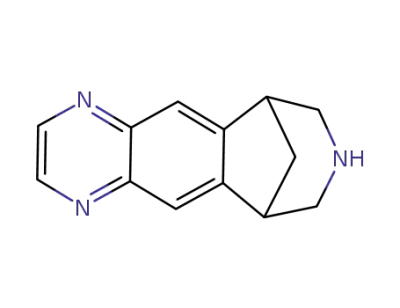
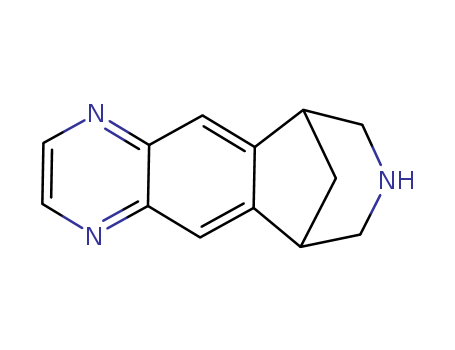
varenicline
| Conditions | Yield |
|---|---|
|
With
palladium 10% on activated carbon; hydrogen;
In
methanol;
for 3h;
under 760.051 Torr;
|
90% |
5,8,14-triazatetracyclo[10.3.1.0(2,11).0(4,9)]hexadeca-2(11),3,5,7,9-pentaene sulfate


varenicline
| Conditions | Yield |
|---|---|
|
With
sodium hydroxide;
In
water;
at 20 - 30 ℃;
for 0.5h;
pH=12.5 - 13.5;
|
83% |
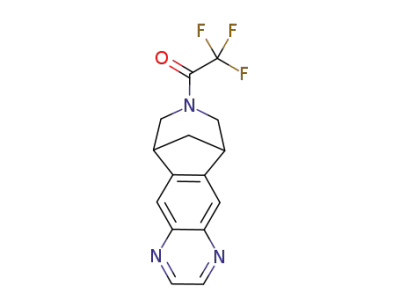
1-(9,10-dihydro-6H-6,10-methanoazepino[4,5-g]quinoxaline-8(7H)-yl)-2,2,2,-trifluoroethanone
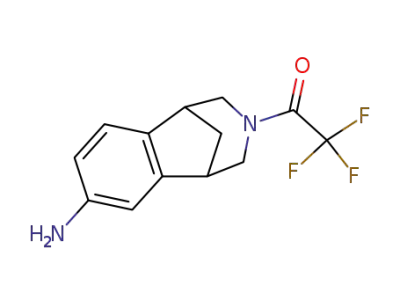
1-(4-amino-10-aza-tricyclo[6.3.1.02,7]dodeca-2,4,6-trien-10-yl)-2,2,2-trifluoro-ethanone
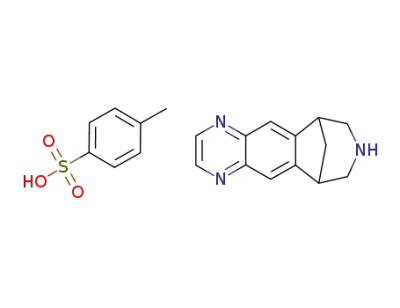
5,8,14-triazatetracyclo[10.3.1.0(2,11).0(4,9)]hexadeca-2(11).3.5,7,9-pentaene tosylate
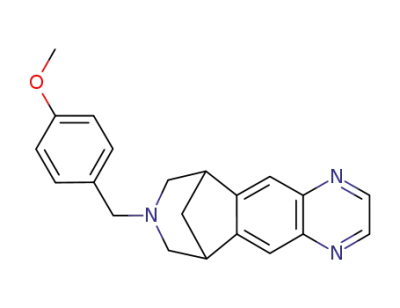
C21H21N3O
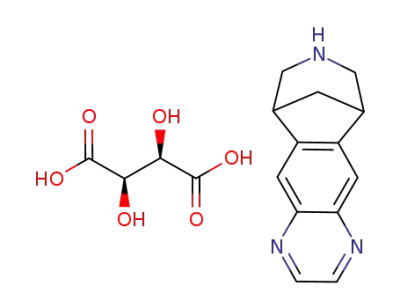
varenicline L-tartarate

5,8,14-triazatetracyclo[10.3.1.0(2,11).0(4,9)]hexadeca-2(11).3.5,7,9-pentaene tosylate
CAS:112163-33-4
CAS:112-84-5
CAS:1477949-42-0
CAS:75225-51-3